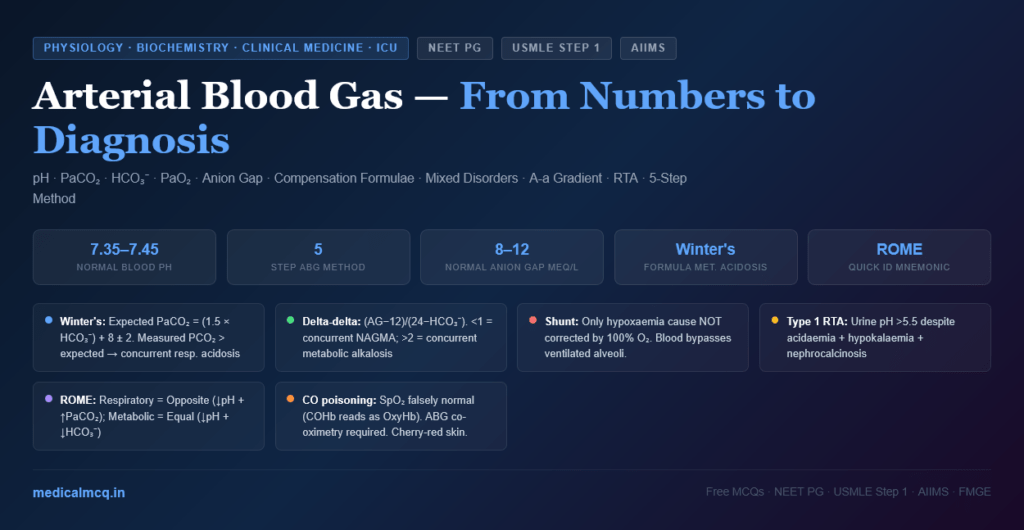
What You Will Learn in This Article
- Understand the physiology of acid-base balance: buffers, lungs, and kidneys
- Define pH, PaCO₂, HCO₃⁻, base excess, PaO₂, and SaO₂ with their normal values
- Apply the Henderson-Hasselbalch equation to acid-base interpretation
- Interpret ABG results using the systematic 5-step approach
- Identify all six primary acid-base disorders with clinical causes
- Calculate and apply compensation formulas for each disorder
- Calculate and interpret the anion gap, osmolal gap, and delta-delta ratio
- Diagnose complex mixed acid-base disorders from clinical scenarios
- Explain the pathophysiology of oxygenation: A-a gradient, P/F ratio, and causes of hypoxaemia
- Apply ABG interpretation to clinical management decisions
📖 Introduction: The Power of ABG Analysis
An unconscious 45-year-old man is brought to the emergency department. His ABG shows: pH 7.10, PaCO₂ 68 mmHg, HCO₃⁻ 20 mEq/L, PaO₂ 52 mmHg, SaO₂ 82%, on room air. Five numbers tell an entire clinical story: this man has combined respiratory and metabolic acidosis, is severely hypoxaemic, and is in immediate danger of respiratory arrest. The ABG guided the clinician to intubate immediately, while subsequent blood investigations revealed diabetic ketoacidosis (metabolic acidosis) superimposed on COPD exacerbation (respiratory acidosis). Without ABG interpretation, neither diagnosis nor the urgency of intervention would have been apparent.
Arterial Blood Gas analysis is one of the most powerful diagnostic tools in clinical medicine — it directly measures what is happening in the blood at the molecular level. For NEET PG and USMLE Step 1, ABG questions test not just memorisation but reasoning: given an ABG, identify the disorder, determine whether compensation is appropriate, calculate the anion gap, and translate the numbers into a clinical diagnosis and management plan.
🔬 Section 1 — Physiology of Acid-Base Balance

1.1 Acids, Bases, and the Concept of pH

Acid: Donates H⁺ ions (proton donor) — H₂CO₃, lactic acid, ketoacids Base: Accepts H⁺ ions (proton acceptor) — HCO₃⁻, Hb, proteins
pH: Negative logarithm of [H⁺] concentration
- pH = −log[H⁺]
- Normal blood pH: 7.35–7.45 (slightly alkaline)
- pH 7.35–7.45 corresponds to [H⁺] = 35–45 nmol/L
- Every 0.1 unit change in pH = ~25% change in [H⁺]
Critical pH values:
- pH <6.8: Incompatible with life (lower limit)
- pH >7.8: Incompatible with life (upper limit)
- pH 7.35–7.45: Normal
- pH <7.35: Acidaemia (excess acid in blood)
- pH >7.45: Alkalaemia (deficit of acid / excess base in blood)
Note on terminology:
- Acidosis = process causing acid accumulation (pH may still be normal if compensated)
- Acidaemia = blood pH <7.35 (the actual state of the blood)
- Alkalosis = process causing base accumulation
- Alkalaemia = blood pH >7.45
1.2 The Three Buffer Systems

Buffers are the first line of defence — act within seconds:
1. Bicarbonate buffer system (most important in ECF): CO₂ + H₂O ⇌ H₂CO₃ ⇌ H⁺ + HCO₃⁻
- Henderson-Hasselbalch equation: pH = pKa + log([HCO₃⁻] / [H₂CO₃])
- Since H₂CO₃ ≈ 0.03 × PaCO₂:
- pH = 6.1 + log([HCO₃⁻] / 0.03 × PaCO₂)
- Normal: pH = 6.1 + log(24 / 0.03 × 40) = 6.1 + log(24 / 1.2) = 6.1 + log(20) = 6.1 + 1.3 = 7.40
- The ratio [HCO₃⁻]:[H₂CO₃] must be 20:1 to maintain normal pH 7.40
2. Phosphate buffer system (most important in urine/ICF): H₂PO₄⁻ ⇌ H⁺ + HPO₄²⁻ (pKa 6.8 — ideal for urine buffering)
3. Protein buffer system (haemoglobin — most important in RBCs):
- Haemoglobin: Most abundant intracellular buffer
- Histidine residues in Hb accept/donate H⁺
- Deoxygenated Hb is a better buffer than oxygenated Hb (Haldane effect — important in CO₂ transport)
1.3 Respiratory Regulation (Second Line — Minutes to Hours)
The respiratory system regulates PaCO₂ (which controls [H₂CO₃]):
CO₂ transport in blood:
- ~5% dissolved in plasma (forms H₂CO₃, measured as PaCO₂)
- ~85% as HCO₃⁻ (formed by carbonic anhydrase in RBCs → HCO₃⁻ exchanges out for Cl⁻ — “chloride shift”)
- ~10% bound to Hb as carbamino compounds
Central chemoreceptors (medulla): Detect [H⁺] in CSF (which reflects arterial PaCO₂) Peripheral chemoreceptors (carotid and aortic bodies): Detect PaO₂ (<60 mmHg triggers response), PaCO₂, and pH
Ventilatory response:
- ↑ PaCO₂ or ↓ pH → ↑ ventilation → ↓ PaCO₂ → raises pH
- ↓ PaCO₂ or ↑ pH → ↓ ventilation → ↑ PaCO₂ → lowers pH
Normal PaCO₂: 35–45 mmHg (respiratory control target)
1.4 Renal Regulation (Third Line — Days)
Kidneys are the primary regulators of HCO₃⁻ concentration — slow but most powerful:
Mechanisms of H⁺ excretion and HCO₃⁻ regulation:
- HCO₃⁻ reabsorption (proximal tubule — 85%):
- Filtered HCO₃⁻ + H⁺ (secreted by NHE3 antiporter) → H₂CO₃ → CO₂ + H₂O
- CO₂ enters tubular cell → carbonic anhydrase → H⁺ + HCO₃⁻ → HCO₃⁻ reabsorbed
- New HCO₃⁻ generation (distal tubule and collecting duct):
- H⁺ secretion into urine via H⁺-ATPase and H⁺/K⁺-ATPase
- H⁺ + HPO₄²⁻ → H₂PO₄⁻ (titratable acid)
- H⁺ + NH₃ → NH₄⁺ (ammonium — major route for H⁺ excretion)
- For every H⁺ excreted, one new HCO₃⁻ is generated and reabsorbed
- Aldosterone: Stimulates H⁺ secretion in collecting duct → generates HCO₃⁻ → promotes alkalosis
Renal response to acidosis: Takes 3–5 days to maximise HCO₃⁻ regeneration Renal response to alkalosis: Takes 3–5 days to maximise HCO₃⁻ excretion
🔬 Section 2 — ABG Parameters: Normal Values and Interpretation
2.1 The ABG Report — What Each Parameter Means
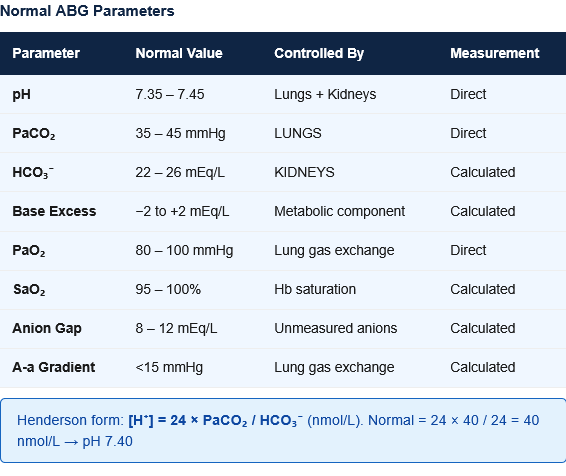
Figure 2: Standard ABG report parameters with normal values. Understanding what each parameter directly measures vs what is calculated is fundamental to correct interpretation.
| Parameter | What It Measures | Normal Value | Units | Directly Measured vs Calculated |
|---|---|---|---|---|
| pH | H⁺ concentration (acidity/alkalinity) | 7.35–7.45 | — | Directly measured |
| PaCO₂ | Partial pressure of CO₂ in arterial blood | 35–45 mmHg | mmHg | Directly measured |
| HCO₃⁻ (actual) | Bicarbonate concentration | 22–26 mEq/L | mEq/L | Calculated from pH + PaCO₂ |
| HCO₃⁻ (standard) | HCO₃⁻ at normal PaCO₂ (40 mmHg) | 22–26 mEq/L | mEq/L | Corrects for respiratory contribution |
| Base Excess (BE) | Amount of acid/base needed to return pH to 7.40 at normal temp + PaCO₂ | −2 to +2 mEq/L | mEq/L | Calculated; negative = base deficit (acidosis) |
| PaO₂ | Partial pressure of O₂ in arterial blood | 80–100 mmHg | mmHg | Directly measured |
| SaO₂ | Percentage of Hb saturated with O₂ | 95–100% | % | Calculated (or measured by co-oximetry) |
| FiO₂ | Fraction of inspired oxygen | 0.21 (room air) | fraction | Set/known |
2.2 Understanding the Henderson Equation (Practical Form)
Instead of the logarithmic Henderson-Hasselbalch, the clinically useful linear form:
[H⁺] (nmol/L) = 24 × PaCO₂ / HCO₃⁻
- Normal: [H⁺] = 24 × 40 / 24 = 40 nmol/L → corresponds to pH 7.40
- Quick relationship: pH 7.40 = [H⁺] 40 nmol/L; each 0.01 pH unit above 7.40 → [H⁺] decreases by ~1 nmol/L
2.3 The Concept of Compensation
Primary disorders always show:
- Primary change: The initiating abnormality (e.g., metabolic acidosis → HCO₃⁻ falls)
- Compensatory response: The body’s attempt to normalize pH by changing the other parameter (e.g., lungs blow off CO₂ → PaCO₂ falls)
Key principles of compensation:
- Compensation NEVER over-corrects — it brings pH towards (not back to) 7.40
- Compensation is predictable — expected ranges can be calculated
- Values outside the expected compensation range → mixed disorder present
- Full compensation = pH returns to normal; only the kidneys can fully compensate respiratory disorders over time (days to weeks)
⚙️ Section 3 — The Systematic 5-Step ABG Interpretation
The Five-Step Method
Example ABG: pH 7.28, PaCO₂ 60 mmHg, HCO₃⁻ 27 mEq/L, PaO₂ 55 mmHg
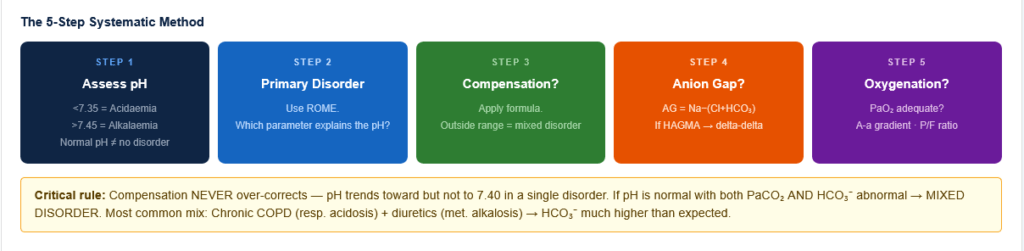
Figure 3: The 5-step systematic ABG interpretation algorithm. Following these steps in order ensures no component of the ABG is missed and mixed disorders are not overlooked.
STEP 1 — Is the patient acidaemic or alkalaemic?
- pH <7.35 → Acidaemia
- pH >7.45 → Alkalaemia
- pH 7.35–7.45 → Normal (but may still have a primary disorder with compensation, or a mixed disorder)
Example: pH 7.28 → Acidaemia
STEP 2 — What is the primary disorder?
Look at PaCO₂ and HCO₃⁻ to identify which is causing the pH abnormality:
| pH | PaCO₂ | HCO₃⁻ | Primary disorder |
|---|---|---|---|
| ↓ | ↑ | ↑ (compensatory) | Respiratory acidosis |
| ↓ | ↓ (compensatory) | ↓ | Metabolic acidosis |
| ↑ | ↓ | ↓ (compensatory) | Respiratory alkalosis |
| ↑ | ↑ (compensatory) | ↑ | Metabolic alkalosis |
Rule: The parameter that EXPLAINS the pH change is the PRIMARY abnormality
- pH ↓ and PaCO₂ ↑ → Respiratory acidosis (high CO₂ causes acidosis)
- pH ↓ and HCO₃⁻ ↓ → Metabolic acidosis (low bicarb causes acidosis)
Example: pH 7.28 (acidaemia), PaCO₂ 60 (high), HCO₃⁻ 27 (high but compensatory) → Primary Respiratory Acidosis
STEP 3 — Is there appropriate compensation?
Calculate the expected compensation using formulas (see Section 4). If the measured value differs significantly from the expected range:
- Measured > Expected → Second primary alkalosis present
- Measured < Expected → Second primary acidosis present
Example: Expected HCO₃⁻ for respiratory acidosis = 24 + (0.35 × ΔPaCO₂) = 24 + (0.35 × 20) = 24 + 7 = 31 mEq/L (acute compensation). Measured HCO₃⁻ = 27. Since acute expected = ~27 and chronic expected = ~31, this is consistent with acute-on-chronic respiratory acidosis.
STEP 4 — Calculate the Anion Gap (if metabolic acidosis present)
Anion Gap (AG) = Na⁺ − (Cl⁻ + HCO₃⁻) Normal AG = 8–12 mEq/L (some labs use 8–16 with older calculation methods)
If AG is elevated (>12), perform additional calculations:
- Delta-delta ratio (ΔAG/ΔHCO₃⁻): = (AG − 12) / (24 − HCO₃⁻)
- 1–2: Pure high-AG metabolic acidosis
- <1: Concurrent normal-AG metabolic acidosis
- 2: Concurrent metabolic alkalosis
STEP 5 — Assess oxygenation
- Is PaO₂ adequate? (Normal 80–100 mmHg on room air)
- Calculate A-a gradient: PAO₂ − PaO₂
- Calculate P/F ratio: PaO₂ / FiO₂
Example: PaO₂ 55 mmHg → Significant hypoxaemia → requires supplemental O₂/ventilatory support
🔬 Section 4 — The Six Primary Acid-Base Disorders
4.1 Respiratory Acidosis

Definition: Primary increase in PaCO₂ (>45 mmHg) → pH falls
Cause: Inadequate ventilation (hypoventilation) → CO₂ retention
Common causes:
- CNS depression: Opioids, benzodiazepines, barbiturates, alcohol, brainstem stroke, head injury, meningitis
- Neuromuscular disease: Guillain-Barré syndrome, myasthenia gravis, motor neurone disease, muscular dystrophy, C3–C5 spinal cord injury (phrenic nerve)
- Airway obstruction: Severe asthma, COPD exacerbation, foreign body, laryngospasm
- Chest wall/lung disease: Flail chest, severe pneumonia, pneumothorax, massive pleural effusion, kyphoscoliosis
- Iatrogenic: Mechanical ventilation with inadequate rate/tidal volume (permissive hypercapnia)
ABG pattern:
- pH ↓, PaCO₂ ↑
- HCO₃⁻: Rises as compensation
Compensation formulae:
| Type | Formula | Time |
|---|---|---|
| Acute respiratory acidosis | Expected HCO₃⁻ = 24 + 0.1 × ΔPCO₂ | Immediate (buffer) |
| Chronic respiratory acidosis | Expected HCO₃⁻ = 24 + 0.35 × ΔPCO₂ | 3–5 days (renal) |
Where: ΔPCO₂ = measured PaCO₂ − 40
Alternative (acute): For every 10 mmHg ↑ PaCO₂ → pH falls by 0.08 (acute) or 0.03 (chronic)
Clinical features:
- Hypercapnia symptoms: Headache (vasodilation → ↑ ICP), confusion, asterixis (CO₂ narcosis)
- Peripheral vasodilation (flushing, bounding pulse)
- Papilloedema (chronic severe hypercapnia)
- CO₂ narcosis → coma (PaCO₂ >80–90 mmHg)
Treatment: Treat underlying cause; improve ventilation; if severe or rapidly worsening → intubation and mechanical ventilation. In COPD: controlled O₂ (target SaO₂ 88–92%) — avoid excessive O₂ which removes hypoxic drive.
4.2 Respiratory Alkalosis

Definition: Primary decrease in PaCO₂ (<35 mmHg) → pH rises
Cause: Hyperventilation → excessive CO₂ elimination
Common causes:
- Hypoxia-driven hyperventilation: High altitude, anaemia, severe pneumonia, PE (early), congenital heart disease
- CNS stimulation: Anxiety/panic attacks, salicylate poisoning (direct stimulation of respiratory centre), stroke, meningitis, encephalitis, fever
- Mechanical hyperventilation: Iatrogenic (ventilator set too fast/deep)
- Liver failure: Hyperammonaemia stimulates respiratory centre
- Pregnancy: Progesterone stimulates respiratory centre (PaCO₂ ~30–32 mmHg normal in pregnancy)
- Sepsis (early): Direct CNS stimulation by cytokines
- Hyperthyroidism
ABG pattern:
- pH ↑, PaCO₂ ↓
- HCO₃⁻: Falls as compensation
Compensation formulae:
| Type | Formula |
|---|---|
| Acute | Expected HCO₃⁻ = 24 − 0.2 × ΔPCO₂ |
| Chronic | Expected HCO₃⁻ = 24 − 0.5 × ΔPCO₂ |
Where: ΔPCO₂ = 40 − measured PaCO₂
Clinical features:
- Dizziness, light-headedness, perioral tingling
- Tetany (hypocalcaemia-like symptoms): ↑ pH → more Ca²⁺ binds to albumin → ↓ ionised Ca²⁺ → Trousseau’s sign, Chvostek’s sign
- Carpopedal spasm
- In panic attacks: self-perpetuating cycle (anxiety → hyperventilation → symptoms → more anxiety)
Treatment: Treat underlying cause. For panic attacks: rebreathing into paper bag (cautiously — not if O₂ concerns), anxiolytics, reassurance, breathing exercises.
4.3 Metabolic Acidosis

Definition: Primary decrease in HCO₃⁻ (<22 mEq/L) → pH falls
Classification — The Anion Gap Framework:
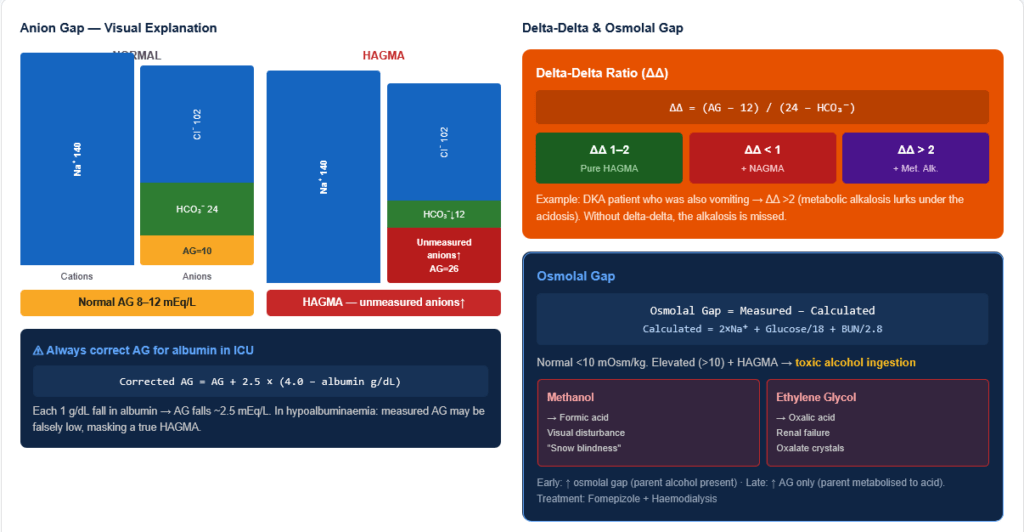
Figure 4: Metabolic acidosis classification by anion gap. High-AG acidosis adds unmeasured anions that consume bicarbonate. Normal-AG (hyperchloraemic) acidosis loses bicarbonate directly or gains chloride. The anion gap is the essential discriminating test.
Anion Gap (AG) = Na⁺ − (Cl⁻ + HCO₃⁻); Normal = 8–12 mEq/L
Correction for hypoalbuminaemia: Corrected AG = measured AG + 2.5 × (4 − measured albumin in g/dL) (Each 1 g/dL fall in albumin reduces normal AG by ~2.5 mEq/L → albumin provides the normal “unmeasured anions”)
High Anion Gap Metabolic Acidosis (HAGMA) — Mnemonic: MUDPILES CAT
| Cause | AG Mechanism | Key Feature |
|---|---|---|
| Methanol | Formic acid accumulation | Visual disturbances (“snow blindness”); ↑ osmolal gap |
| Uraemia (renal failure) | Organic acids accumulate | BUN ↑, Creatinine ↑; chronic CKD |
| Diabetic ketoacidosis (DKA) | Acetoacetate + β-hydroxybutyrate | Glucose ↑, ketones ↑; type 1 DM |
| Propylene glycol | Lactic acid metabolite | IV lorazepam/other medications |
| Isoniazid / Iron / Inborn errors | Lactic acid (isoniazid inhibits IDH) | History of TB treatment |
| Lactic acidosis | Lactate accumulation | Type A: hypoperfusion; Type B: metformin, cyanide |
| Ethylene glycol | Oxalic acid → renal failure | Antifreeze ingestion; ↑ osmolal gap, oxalate crystals in urine |
| Salicylates | Direct + respiratory alkalosis | Dual disorder; tinnitus |
| CO poisoning | Lactic acidosis (tissue hypoxia) | SaO₂ normal on pulse oximetry; SpCO elevated |
| Alcoholic ketoacidosis | Ketones (low or normal glucose) | Binge + starvation; glucose normal/low |
| Toluene / Thyroid storm | Metabolic acid | Glue-sniffing; tachycardia + hyperthermia |
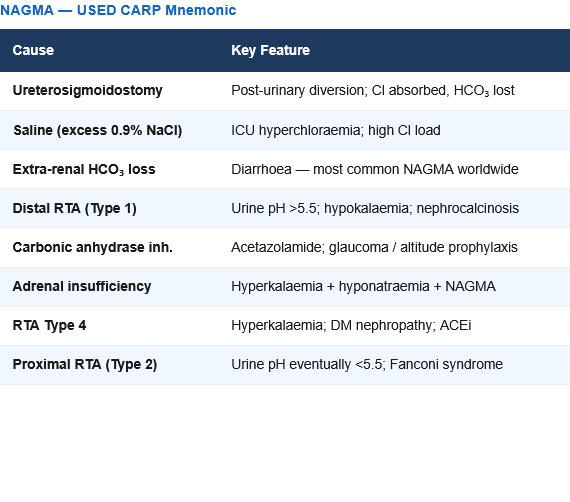
Normal Anion Gap Metabolic Acidosis (NAGMA) — Hyperchloraemic: Mnemonic: USED CARP
| Cause | Mechanism | Key Feature |
|---|---|---|
| Ureteroenteric fistula | Intestine absorbs Cl⁻ excretes HCO₃⁻ | Post-urinary diversion surgery |
| Saline infusion (excessive 0.9% NaCl) | Dilutional / hyperchloraemic | ICU patients; high Cl⁻ load |
| ExtraRenal HCO₃⁻ loss (diarrhoea) | Direct HCO₃⁻ loss in stool | Most common cause of NAGMA worldwide |
| Distal RTA (type 1) | Cannot excrete H⁺ → cannot regenerate HCO₃⁻ | Urine pH >5.5 despite acidaemia; nephrocalcinosis; hypokalaemia |
| Carbonic anhydrase inhibitors | Acetazolamide blocks HCO₃⁻ reabsorption → HCO₃⁻ lost in urine | Glaucoma treatment; altitude sickness prophylaxis |
| Adrenal insufficiency | ↓ Aldosterone → ↓ H⁺ excretion | Hyperkalaemia + hyponatraemia + NAGMA |
| Renal tubular acidosis Type 4 | Hyperkalaemia → impaired NH₄⁺ excretion | Diabetic nephropathy; hyperkalaemia |
| Proximal RTA (type 2) | Cannot reabsorb filtered HCO₃⁻ | Urine pH <5.5; Fanconi syndrome |
Compensation formula:
- Winter’s formula: Expected PaCO₂ = (1.5 × HCO₃⁻) + 8 ± 2
- Alternatively: Expected PaCO₂ ≈ HCO₃⁻ + 15 (simpler mnemonic)
- If measured PaCO₂ > expected → concurrent respiratory acidosis
- If measured PaCO₂ < expected → concurrent respiratory alkalosis
ABG pattern: pH ↓, HCO₃⁻ ↓, PaCO₂ ↓ (compensatory hyperventilation — Kussmaul breathing in severe DKA)
Clinical features: Kussmaul respiration (deep, rapid — compensatory), nausea/vomiting, confusion, fatigue; specific features depend on cause (fruity breath in DKA, tinnitus in salicylate, visual symptoms in methanol)
4.4 Metabolic Alkalosis

Definition: Primary increase in HCO₃⁻ (>26 mEq/L) → pH rises
Causes — Two categories:
Generation of alkalosis (how it starts):
- Vomiting or nasogastric suction (loss of HCl → loss of H⁺ → HCO₃⁻ rises)
- Antacid use, alkali ingestion (NaHCO₃)
- Diuretics (loop/thiazides → ↑ distal H⁺ secretion, ↑ aldosterone → “contraction alkalosis”)
- Hypokalaemia → K⁺ exits cells, H⁺ enters cells → intracellular acidosis → kidney secretes H⁺ → ↑ HCO₃⁻
- Primary hyperaldosteronism → ↑ H⁺ secretion in collecting duct
Maintenance of alkalosis (why it persists — kidney normally excretes excess HCO₃⁻):
- Volume depletion (hypovolaemia → avid Na⁺ reabsorption → HCO₃⁻ co-absorbed) → “saline-responsive” alkalosis
- Hypokalaemia → kidney continues to retain HCO₃⁻ (exchanges K⁺ for H⁺ → more H⁺ secreted)
- Chloride depletion → HCO₃⁻ must be retained to maintain electrochemical balance
- Hyperaldosteronism → aldosterone drives H⁺ secretion → “saline-resistant” alkalosis
Saline-responsive vs Saline-resistant:
| Feature | Saline-Responsive | Saline-Resistant |
|---|---|---|
| Urine Cl⁻ | <25 mEq/L (Cl⁻-avid state) | >40 mEq/L |
| Causes | Vomiting, NG suction, diuretics (remote), post-hypercapnia | Hyperaldosteronism, Cushing’s, Bartter/Gitelman syndrome, severe hypokalaemia |
| Treatment | IV normal saline + KCl | Treat underlying cause; spironolactone |
Compensation formula:
- Expected PaCO₂ = 40 + 0.7 × (HCO₃⁻ − 24) [or: 0.6–0.7 × ΔHCO₃⁻]
- Maximum compensation: PaCO₂ rarely rises above 55–60 mmHg (hypoxia limits hypoventilation)
ABG pattern: pH ↑, HCO₃⁻ ↑, PaCO₂ ↑ (compensatory hypoventilation)
Clinical features: Nausea, weakness, muscle cramps; if severe → cardiac arrhythmias, decreased mental status; tetany (from decreased ionised Ca²⁺ if pH very high)
🔬 Section 5 — Compensation Formulae: Complete Reference
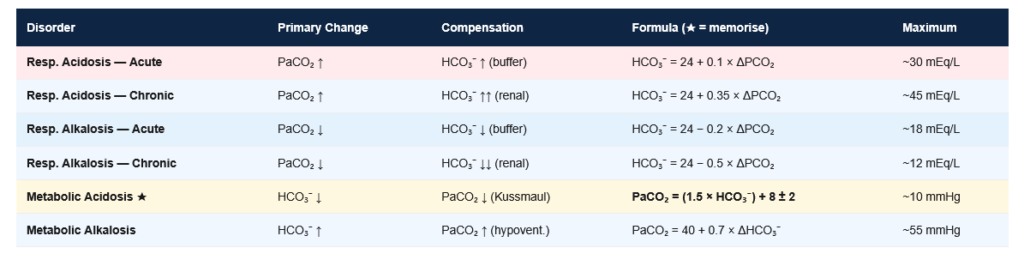
Figure 5: Complete acid-base compensation formulae reference. Memorise these formulae — exam questions frequently present an ABG and ask whether the compensation is appropriate (consistent with a single primary disorder) or whether a mixed disorder is present.
| Disorder | Primary Change | Compensation | Formula | Max Compensation |
|---|---|---|---|---|
| Respiratory Acidosis (Acute) | PaCO₂ ↑ | HCO₃⁻ ↑ | HCO₃⁻ = 24 + 0.1 × ΔPCO₂ | ~30 mEq/L |
| Respiratory Acidosis (Chronic) | PaCO₂ ↑ | HCO₃⁻ ↑↑ | HCO₃⁻ = 24 + 0.35 × ΔPCO₂ | ~45 mEq/L |
| Respiratory Alkalosis (Acute) | PaCO₂ ↓ | HCO₃⁻ ↓ | HCO₃⁻ = 24 − 0.2 × ΔPCO₂ | ~18 mEq/L |
| Respiratory Alkalosis (Chronic) | PaCO₂ ↓ | HCO₃⁻ ↓↓ | HCO₃⁻ = 24 − 0.5 × ΔPCO₂ | ~12–15 mEq/L |
| Metabolic Acidosis | HCO₃⁻ ↓ | PaCO₂ ↓ | PaCO₂ = (1.5 × HCO₃⁻) + 8 ± 2 (Winter’s formula) | ~10–12 mmHg |
| Metabolic Alkalosis | HCO₃⁻ ↑ | PaCO₂ ↑ | PaCO₂ = 40 + 0.7 × ΔHCO₃⁻ | ~55–60 mmHg |
ΔPCO₂ = |measured PaCO₂ − 40| ΔHCO₃⁻ = |measured HCO₃⁻ − 24|
Quick rules for the exam:
- For metabolic acidosis: “PCO₂ should equal HCO₃⁻ + 15” (simpler than Winter’s — approximate but useful)
- For respiratory acidosis acute: Each 10 mmHg ↑ PaCO₂ → HCO₃⁻ rises by 1 mEq/L
- For respiratory acidosis chronic: Each 10 mmHg ↑ PaCO₂ → HCO₃⁻ rises by 3.5 mEq/L
- For respiratory alkalosis acute: Each 10 mmHg ↓ PaCO₂ → HCO₃⁻ falls by 2 mEq/L
- For respiratory alkalosis chronic: Each 10 mmHg ↓ PaCO₂ → HCO₃⁻ falls by 5 mEq/L
🔬 Section 6 — Anion Gap, Osmolal Gap, and Delta-Delta
6.1 Anion Gap (AG)
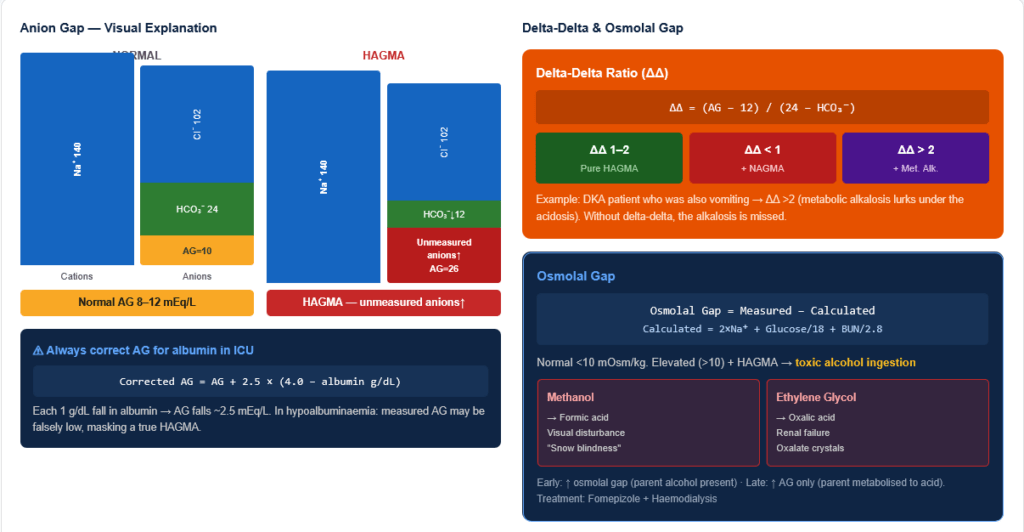
AG = Na⁺ − (Cl⁻ + HCO₃⁻) → Normal 8–12 mEq/L (some use 10–12 with albumin correction)
Why does a gap exist? Blood is electroneutral but not all anions are measured. Unmeasured anions (albumin ~2 g/dL contributes ~12 mEq/L, phosphate, sulphate, organic anions) are larger than unmeasured cations (K⁺, Ca²⁺, Mg²⁺ partly) → apparent “gap.”
AG increases when: Unmeasured anions accumulate (lactate, ketoacids, uraemic acids, toxin metabolites) AG decreases when: Albumin falls (each 1 g/dL fall → AG falls by ~2.5) OR unmeasured cations rise (multiple myeloma with cationic Ig, lithium, calcium)
Always correct AG for albumin in critically ill patients: Corrected AG = measured AG + 2.5 × (4.0 − albumin g/dL)
6.2 Osmolal Gap
Measured osmolality − Calculated osmolality
Calculated osmolality = 2 × Na⁺ + Glucose/18 + BUN/2.8 (in mg/dL units) Or: = 2 × Na⁺ + Glucose (mmol/L) + Urea (mmol/L) in SI units
Normal osmolal gap: <10 mOsm/kg
Elevated osmolal gap (>10) + HAGMA → Suggests toxic alcohol ingestion:
- Methanol (wood alcohol — metabolised to formic acid; optic neuropathy, snow-blindness)
- Ethylene glycol (antifreeze — metabolised to oxalic acid; renal failure; oxalate crystals in urine)
- Propylene glycol (medication solvent)
- Ethanol itself elevates osmolal gap but not AG
The sequence with toxic alcohols:
- Early: ↑ osmolal gap (parent alcohol present) + normal or ↑ AG
- Late: ↓ osmolal gap (parent metabolised) + ↑ AG (toxic metabolites)
6.3 Delta-Delta Ratio (ΔΔ)
When: Used to detect a concurrent second metabolic disorder in a patient with HAGMA
Formula: ΔΔ = (AG − 12) / (24 − HCO₃⁻) = change in AG / change in HCO₃⁻
Interpretation:
- ΔΔ = 1–2: Pure HAGMA (the rise in AG exactly matches the fall in HCO₃⁻)
- ΔΔ < 1: Concurrent NAGMA (HCO₃⁻ has fallen MORE than the AG has risen → additional HCO₃⁻ loss from another source)
- ΔΔ > 2: Concurrent metabolic alkalosis (HCO₃⁻ is higher than expected for the AG elevation → a baseline alkalosis was present)
Clinical example: A patient with vomiting (metabolic alkalosis) then develops DKA (HAGMA). The metabolic alkalosis had raised HCO₃⁻ to 35. Now DKA drops HCO₃⁻ by 12 (to 23 — which is “normal”). But the AG is elevated by 12. The ΔΔ = 12 / 1 = 12 → indicates a concurrent metabolic alkalosis that was present before the DKA. Without delta-delta, you would miss the alkalosis.
🔬 Section 7 — Renal Tubular Acidosis (RTA): NAGMA in Detail
7.1 Overview and Urine Anion Gap
When a patient has NAGMA (normal anion gap metabolic acidosis), the key is to determine whether the kidneys are the cause or the gut is the cause:
Urine Anion Gap (UAG) = Urine Na⁺ + Urine K⁺ − Urine Cl⁻
- Normal/+ve UAG (>0): NH₄⁺ excretion is LOW → kidney is FAILING to excrete acid → Renal cause (RTA)
- Negative UAG (<0): NH₄⁺ excretion is HIGH → kidneys are working properly → Extrarenal cause (diarrhoea)
Why? NH₄⁺ is unmeasured (like Na/K but not Cl) → when NH₄⁺ is high in urine (kidneys responding appropriately), Cl⁻ must also be high (for electrochemical balance) → Cl⁻ > Na⁺ + K⁺ → UAG goes negative. Conversely, when NH₄⁺ is low (kidney failing to respond), Cl⁻ is relatively low → Na⁺ + K⁺ > Cl⁻ → UAG is positive.
7.2 RTA Types Comparison
Figure 6: Renal tubular acidosis — comparison of three types. The urine pH response to systemic acidaemia is the key distinguishing test: Type 1 cannot lower urine pH below 5.5; Type 2 eventually can; Type 4 is characterised by hyperkalaemia rather than hypokalaemia.
| Feature | Type 1 (Distal) | Type 2 (Proximal) | Type 4 (Hyperkalaemic) |
|---|---|---|---|
| Defect | Cannot secrete H⁺ in collecting duct | Cannot reabsorb HCO₃⁻ in proximal tubule | ↓ Aldosterone → ↓ H⁺ + K⁺ secretion |
| Serum K⁺ | ↓ (hypo) | ↓ (hypo) | ↑ (hyper) |
| Urine pH | >5.5 (cannot acidify) | <5.5 (distal can compensate eventually) | <5.5 |
| HCO₃⁻ threshold | Normal | Low (wastes HCO₃⁻ easily) | Low-normal |
| Causes | Sjögren’s, RA, SLE, amphotericin B, hereditary (CAI mutation) | Fanconi syndrome, multiple myeloma, Wilson’s, carbonic anhydrase inhibitors | Diabetes (most common), hypoaldosteronism, ACE inhibitors, spironolactone, CKD |
| Complications | Nephrocalcinosis, nephrolithiasis (CaOx/CaPO₄) | Rickets, osteomalacia (phosphate wasting) | Hyperkalaemia complications |
| Treatment | Alkali supplementation (NaHCO₃, K citrate) | Treat underlying cause; high-dose alkali needed | Fludrocortisone; treat cause |
Type 3 RTA: Combined Type 1 + Type 2 (very rare; carbonic anhydrase II deficiency; autosomal recessive) No Type 5: Classification skips to Type 4 (no “Type 3” in most modern classifications)
🔬 Section 8 — Oxygenation: PaO₂, A-a Gradient, and P/F Ratio
8.1 PaO₂ and Hypoxaemia
Normal PaO₂: 80–100 mmHg on room air (FiO₂ 0.21, sea level) Rule of thumb: Expected PaO₂ ≈ FiO₂ × 500 (on mechanical ventilation)
Hypoxaemia classification by severity:
- Mild: PaO₂ 60–80 mmHg
- Moderate: PaO₂ 40–60 mmHg
- Severe: PaO₂ <40 mmHg
Effect of age on PaO₂: Expected PaO₂ (mmHg) = 100 − (0.3 × age in years) [or 100 − age/3]
8.2 The Alveolar Gas Equation and A-a Gradient
Alveolar Gas Equation: PAO₂ = (FiO₂ × [Pₐₜₘ − PH₂O]) − (PaCO₂ / RQ)
Simplified: PAO₂ = (FiO₂ × 713) − PaCO₂/0.8 on room air at sea level: PAO₂ = (0.21 × 713) − PaCO₂/0.8 = 149.7 − PaCO₂/0.8
At sea level: Pₐₜₘ = 760 mmHg; PH₂O = 47 mmHg; RQ = 0.8
A-a gradient (Alveolar-arterial oxygen gradient): A-a gradient = PAO₂ − PaO₂
Normal A-a gradient:
- Young adult (~20 years): ~5–10 mmHg
- Increases with age: 2.5 + (0.21 × age) [approximately]
- On 100% O₂: Normal <100 mmHg
Causes of hypoxaemia — using A-a gradient:
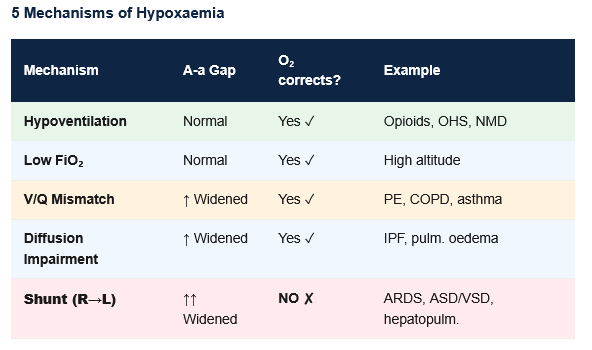
Figure 7: Five mechanisms of hypoxaemia and their A-a gradient. The A-a gradient is the essential test: a normal gradient with hypoxaemia means the lungs are normal (hypoventilation or low FiO₂). A widened gradient indicates intrinsic lung or vascular pathology.
| Mechanism | A-a Gradient | PaO₂ response to O₂ | Clinical Examples |
|---|---|---|---|
| Hypoventilation | Normal | Improves | Opioid overdose, obesity hypoventilation, neuromuscular disease |
| V/Q Mismatch | ↑ Widened | Improves | PE, COPD, asthma, atelectasis, mucus plugging |
| Diffusion Impairment | ↑ Widened | Improves | Interstitial lung disease (IPF, sarcoidosis), pulmonary oedema (mild) |
| Shunt (R→L) | ↑ Widened | Does NOT improve significantly | ARDS, congenital heart disease (ASD, VSD), hepatopulmonary syndrome, severe pneumonia |
| Low FiO₂ | Normal | Improves | High altitude, closed space O₂ depletion |
Shunt distinction: 100% O₂ does NOT correct shunt hypoxaemia — blood passing through non-ventilated areas mixes with oxygenated blood and significantly lowers PaO₂. This is the diagnostic hallmark of true shunting.
8.3 P/F Ratio (Horowitz Index)
P/F ratio = PaO₂ / FiO₂
Normal P/F ratio: >400 mmHg (on room air: 95 / 0.21 ≈ 452)
Clinical use — Berlin criteria for ARDS:
| ARDS severity | P/F ratio | Clinical significance |
|---|---|---|
| Mild ARDS | 200–300 | With PEEP/CPAP ≥5 cm H₂O |
| Moderate ARDS | 100–200 | With PEEP ≥5 |
| Severe ARDS | <100 | With PEEP ≥5 |
Non-ARDS use:
- P/F <300 → significant hypoxaemia
- P/F <200 → ARDS range; usually requires mechanical ventilation
- P/F ratio quick calculation: If on 40% O₂ (FiO₂ 0.4) and PaO₂ is 80 → P/F = 200 (moderate ARDS)
🏥 Section 9 — Complex ABG Scenarios: Mixed Disorders
9.1 Recognising Mixed Disorders
A mixed acid-base disorder exists when two or more primary disorders occur simultaneously. Clues:
- pH is normal (7.35–7.45) despite both PaCO₂ and HCO₃⁻ being abnormal
- Compensation is excessive or insufficient compared to expected
- Delta-delta ratio is <1 or >2
9.2 Common Mixed Disorders
Mixed Respiratory + Metabolic Acidosis:
- pH ↓↓ (severely low), PaCO₂ ↑, HCO₃⁻ ↓ (both pulling pH down in same direction)
- No compensation — both are primary disorders, both worsening pH
- Example: Cardiac arrest (respiratory failure + lactic acidosis), COPD patient with DKA, drowning
Mixed Respiratory + Metabolic Alkalosis:
- pH ↑↑ (very high), PaCO₂ ↓, HCO₃⁻ ↑ (both pulling pH up)
- Example: Vomiting + mechanical hyperventilation, primary hyperaldosteronism + anxiety, liver failure with diuretics
Metabolic Acidosis + Metabolic Alkalosis:
- pH may be normal; HCO₃⁻ near normal but AG is elevated
- Delta-delta >2 (alkalosis is present “under” the acidosis)
- Example: DKA + vomiting; uraemia + vomiting; HFI with diuretics
Respiratory Acidosis + Metabolic Alkalosis (most common mixed disorder in clinical practice!):
- pH may be near normal; PaCO₂ ↑; HCO₃⁻ ↑↑ (more than expected for chronic resp. acidosis)
- Example: COPD patient on diuretics (diuretics cause metabolic alkalosis on top of chronic hypercapnia)
- Clue: HCO₃⁻ > expected for the degree of PaCO₂ elevation
Triple acid-base disorder:
- Three simultaneous disorders (e.g., chronic respiratory acidosis + metabolic alkalosis + HAGMA in a critically ill COPD patient with DKA)
- Requires systematic analysis of all parameters
9.3 Worked ABG Examples
Example 1: pH 7.10, PaCO₂ 20 mmHg, HCO₃⁻ 6 mEq/L, Na⁺ 140, Cl⁻ 110, glucose 450 mg/dL
Step 1: pH 7.10 → Acidaemia Step 2: HCO₃⁻ ↓ (primary driver) → Metabolic acidosis Step 3: Expected PaCO₂ = (1.5 × 6) + 8 ± 2 = 17 ± 2 → Measured 20 → Slightly high → Possibly concurrent respiratory acidosis but within range Step 4: AG = 140 − (110 + 6) = 24 → High-AG metabolic acidosis; Glucose 450 → DKA Step 5: Likely hypoxaemia (not given) — assess PaO₂
Conclusion: Diabetic Ketoacidosis (HAGMA) with compensatory respiratory alkalosis (Kussmaul breathing)
Example 2: pH 7.44, PaCO₂ 55 mmHg, HCO₃⁻ 37 mEq/L
Step 1: pH 7.44 → Normal/borderline alkalosis Step 2: PaCO₂ 55 (↑) + HCO₃⁻ 37 (↑) — both elevated. If this were simple chronic respiratory acidosis: expected HCO₃⁻ = 24 + 0.35 × 15 = 29.25. Measured HCO₃⁻ = 37 >> expected 29. Therefore HCO₃⁻ is too high for the PaCO₂ elevation. Step 3: Concurrent metabolic alkalosis present (HCO₃⁻ too high)
Conclusion: Chronic respiratory acidosis (COPD) + Metabolic alkalosis (likely from diuretics). Most common mixed disorder in clinical practice.
🔬 Section 10 — Special Situations in ABG
10.1 ABG in Pregnancy
Normal ABG changes in pregnancy:
- PaCO₂: ↓ to ~30–32 mmHg (progesterone → respiratory stimulation → hyperventilation)
- HCO₃⁻: ↓ to ~18–20 mEq/L (compensatory renal HCO₃⁻ excretion)
- pH: Slightly elevated (~7.44)
- PaO₂: Slightly elevated on room air
Key implication: In pregnancy, a “normal” PaCO₂ of 40 mmHg represents hypoventilation (the expected is 30–32 mmHg) and may indicate impending respiratory failure.
10.2 ABG in High Altitude
- ↓ FiO₂ (same 21% but lower atmospheric pressure) → ↓ PAO₂ → Hypoxia
- Hypoxia → Hyperventilation → ↓ PaCO₂ → Respiratory alkalosis
- Compensation: Kidneys excrete HCO₃⁻ over days → HCO₃⁻ falls → pH normalises
- Acetazolamide (carbonic anhydrase inhibitor) accelerates HCO₃⁻ excretion → speeds up acclimatisation
10.3 Venous Blood Gas (VBG) vs ABG
- VBG pH is ~0.03–0.05 lower than ABG pH (acceptable for screening)
- VBG PaCO₂ is ~5–6 mmHg higher than ABG
- VBG HCO₃⁻ is ~1–2 mEq/L higher
- VBG PaO₂ is NOT reliable for oxygenation assessment
- Use: VBG is adequate for acid-base assessment when arterial access is difficult; NOT for oxygenation
10.4 Pulse Oximetry vs ABG SaO₂
- Pulse oximetry (SpO₂): Non-invasive; measures oxyhaemoglobin vs deoxyhaemoglobin
- Limitations:
- Cannot detect carboxyhaemoglobin (CO poisoning — SpO₂ reads falsely normal)
- Cannot detect methaemoglobin (reads ~85% regardless)
- Inaccurate when SaO₂ <80%, poor perfusion, dark pigmentation, nail polish
- Only measures O₂ saturation — NOT PaO₂, pH, or CO₂
- ABG co-oximetry can directly measure carboxyHb, metHb, and other Hb fractions
🔄 Section 11 — Connections to Other Topics
→ Renal Physiology — The kidneys are the primary long-term regulators of acid-base balance through HCO₃⁻ reabsorption (proximal tubule), H⁺ secretion, and NH₄⁺ excretion. CKD causes NAGMA (uraemia → HAGMA in late stages). Aldosterone drives H⁺ excretion in the collecting duct — its excess causes metabolic alkalosis; its deficiency causes Type 4 RTA.
→ Respiratory Physiology — The ventilatory response to acidosis is the fastest compensation mechanism (minutes). Understanding V/Q relationships, dead space, shunt, and alveolar gas equation links ABG directly to pulmonary pathology. ARDS management is guided by P/F ratio and ABG-based ventilator settings.
→ Electrolytes — Potassium and acid-base balance are intimately linked. Acidosis → K⁺ exits cells (hyperkalaemia); alkalosis → K⁺ enters cells (hypokalaemia). Hypokalaemia causes and maintains metabolic alkalosis. Hyperkalaemia is the hallmark of Type 4 RTA. Never correct alkalaemia without also correcting hypokalaemia.
→ Diabetic Emergencies — DKA produces HAGMA (ketoacids) with compensatory hyperventilation (Kussmaul breathing). HHS (Hyperosmolar Hyperglycaemic State) shows osmolal gap but milder or no acidosis. DKA management requires serial ABGs to monitor resolution of acidosis and avoid overtreatment (rebound metabolic alkalosis from bicarb).
→ Toxicology — Salicylate poisoning classically produces a mixed respiratory alkalosis + metabolic acidosis (early alkalosis from direct stimulation, later acidosis from salicylate’s metabolic effects). Methanol and ethylene glycol → HAGMA + elevated osmolal gap. CO poisoning → lactic acidosis with normal pulse oximetry (SpO₂). Understanding ABG in poisoning is essential for toxicological management.

🎯 High-Yield Exam Facts
🔴 Winter’s formula: Expected PaCO₂ in metabolic acidosis = (1.5 × HCO₃⁻) + 8 ± 2 If measured PaCO₂ is higher than expected → concurrent respiratory acidosis. If lower → concurrent respiratory alkalosis. This formula must be memorised for exam ABG interpretation.
🔴 In chronic respiratory acidosis, HCO₃⁻ rises by 3.5 mEq/L for every 10 mmHg rise in PaCO₂ (renal compensation over 3–5 days) In acute: only 1 mEq/L per 10 mmHg (buffer response only). The distinction between acute and chronic is determined by the degree of HCO₃⁻ compensation.
🔴 Delta-delta ratio < 1 → concurrent NAGMA; > 2 → concurrent metabolic alkalosis Delta-delta = (AG − 12) / (24 − HCO₃⁻). Always calculate this in HAGMA. A DKA patient with vomiting has ΔΔ >2. A DKA patient with diarrhoea has ΔΔ <1.
🔴 Type 1 RTA (Distal): urine pH >5.5 despite systemic acidaemia; HYPOKALAEMIA; nephrocalcinosis Cannot acidify urine because H⁺-ATPase is defective. K⁺ wasted instead of H⁺. Calcium phosphate stones/nephrocalcinosis because high urine pH precipitates CaHPO₄.
🔴 Shunt: only cause of hypoxaemia that does NOT correct with 100% O₂ V/Q mismatch, diffusion impairment, and hypoventilation all improve with supplemental O₂. Shunt (blood bypassing ventilated alveoli) does not — the shunted blood continues to mix with oxygenated blood, limited by cardiac output physics.
🔴 COPD + Diuretics = most common mixed acid-base disorder (Respiratory acidosis + Metabolic alkalosis) HCO₃⁻ will be much higher than expected for chronic respiratory acidosis alone. pH may be normal. Identify this by checking if HCO₃⁻ exceeds the chronic compensation formula result.
🟠 Urine anion gap (UAG) distinguishes RTA from diarrhoea in NAGMA UAG = UNa⁺ + UK⁺ − UCl⁻. Negative UAG (Cl⁻ > Na⁺ + K⁺) → NH₄⁺ high → kidneys responding → extrarenal cause (diarrhoea). Positive UAG → NH₄⁺ low → kidney is not acidifying properly → RTA.
🟠 Metabolic alkalosis is maintained by chloride depletion and hypokalaemia — treat with IV NaCl + KCl Urine Cl⁻ <25 mEq/L → saline-responsive (vomiting, diuretics). Urine Cl⁻ >40 mEq/L → saline-resistant (hyperaldosteronism, severe hypokalaemia).
🟠 Pregnancy: “Normal” PaCO₂ 40 mmHg represents hypoventilation — expected is 30–32 mmHg Always apply pregnancy-specific ABG norms. HCO₃⁻ is also normally low (18–20 mEq/L) in pregnancy. A pregnant woman with PaCO₂ 40 may be in impending respiratory failure.
🟠 Osmolal gap >10 + HAGMA → toxic alcohol (methanol or ethylene glycol) Methanol → formic acid (visual symptoms — “snow blindness”). Ethylene glycol → oxalic acid (renal failure, oxalate crystals in urine). Early: high osmolal gap. Late: high AG (toxin metabolised), oxalate crystals. Treatment: fomepizole (blocks alcohol dehydrogenase) or ethanol (competitive substrate).
🟡 Salicylate poisoning = mixed respiratory alkalosis + metabolic acidosis pH may be near-normal despite both disorders. Respiratory alkalosis (direct medullary stimulation) appears first. Then metabolic acidosis (salicylate uncouples oxidative phosphorylation → lactic acid, ketoacids). Treatment: alkaline diuresis (urinary pH >7.5 → traps ionised salicylate), haemodialysis if severe.
🟡 A-a gradient increases with age and with elevated FiO₂ Normal young adult: ~5–10 mmHg. Rule: A-a = 2.5 + (0.21 × age). On 100% O₂: normal A-a <100 mmHg. On room air, elevated A-a = intrinsic pulmonary cause of hypoxaemia.
🟡 In Type 4 RTA, the hallmark is hyperkalaemia — this is how it differs from Types 1 and 2 Caused by hypoaldosteronism (Addison’s, diabetic nephropathy, ACE inhibitors, NSAIDs, trimethoprim). The hyperkalaemia itself impairs NH₄⁺ production → failure to excrete H⁺ → NAGMA + hyperkalaemia.
🧠 Mnemonics
“MUDPILES CAT” — HAGMA causes Methanol · Uraemia · DKA · Propylene glycol · Isoniazid/Iron/Inborn errors · Lactic acidosis · Ethylene glycol · Salicylates · CO poisoning · Alcoholic ketoacidosis · Toluene/Thyroid storm
“USED CARP” — NAGMA causes (Normal AG / Hyperchloraemic) Ureteroenterostomy · Saline (excessive) · ExtraRenal HCO₃⁻ loss (diarrhoea) · Distal RTA (Type 1) · Carbonic anhydrase inhibitors · Adrenal insufficiency · RTA Type 4 · Proximal RTA (Type 2)
“RomanS GIVES HiCO₃” — Metabolic Alkalosis causes Vomiting / NG suction → Gives HCO₃⁻ (removes H⁺) Iatrogenic alkali Diuretics → loss of Cl⁻ / volume → contraction alkalosis Hyperaldosteronism → H⁺ secretion in CD
“ROME” — Quick primary disorder identification Respiratory: Opposite (pH and PaCO₂ move in OPPOSITE directions: pH ↓ + PaCO₂ ↑) Metabolic: Equal (pH and HCO₃⁻ move in the SAME direction: pH ↓ + HCO₃⁻ ↓)
“PCO₂ = HCO₃⁻ + 15” — Quick Winter’s check For metabolic acidosis: PaCO₂ should approximately equal HCO₃⁻ + 15. If PaCO₂ = 20 and HCO₃⁻ = 10 → 10 + 15 = 25, but measured = 20 → close to expected (some respiratory compensation present).
⚠️ Common Mistakes
❌ “Normal pH means no acid-base disorder” ✅ A mixed disorder (e.g., respiratory acidosis + metabolic alkalosis) can result in a normal pH. Always check PaCO₂ and HCO₃⁻ even if pH is 7.40. If both are elevated or both are low, a mixed disorder is present.
❌ “If compensation brings pH to normal, there are no problems” ✅ Compensation never over-corrects a primary disorder in simple (unmixed) cases. If pH is NORMAL with abnormal PaCO₂ and HCO₃⁻, it implies a mixed disorder, not perfect compensation of a single disorder.
❌ “Metabolic alkalosis can compensate fully by holding breath” ✅ Hypoventilation to compensate metabolic alkalosis has a limit — the resulting hypoxia stimulates ventilation, preventing PaCO₂ from rising above ~55–60 mmHg in compensation. This is the maximum compensation limit for metabolic alkalosis.
❌ “RTA Type 1 and Type 2 are distinguished only by their number” ✅ Key distinction: Type 1 (Distal) → urine pH >5.5 (cannot acidify), hypokalaemia, nephrocalcinosis. Type 2 (Proximal) → urine pH <5.5 (can acidify distally when bicarbonate threshold is exceeded), hypokalaemia, part of Fanconi syndrome (aminoaciduria + glycosuria + phosphaturia). Type 4 is unique: hyperkalaemia + low urine pH.
❌ “DKA beta-hydroxybutyrate will be detected by urine ketone dipstick” ✅ Standard urine dipsticks (nitroprusside reaction) detect ACETOACETATE and acetone — NOT beta-hydroxybutyrate (the predominant ketone in DKA). In early DKA, beta-hydroxybutyrate predominates and dipstick may be falsely low. As treatment proceeds, beta-hydroxybutyrate converts to acetoacetate → dipstick may paradoxically “worsen” despite clinical improvement. Use serum beta-hydroxybutyrate for monitoring.
📝 5 Practice MCQs
Q1: A 68-year-old man with COPD is brought in with worsening breathlessness. ABG: pH 7.30, PaCO₂ 72 mmHg, HCO₃⁻ 34 mEq/L. He is known to have had PaCO₂ 55 mmHg on a baseline outpatient ABG. What is the correct interpretation?
- A. Acute respiratory acidosis with appropriate compensation
- B. Chronic respiratory acidosis with appropriate compensation
- C. Acute-on-chronic respiratory acidosis — HCO₃⁻ below expected for purely acute change
- D. Mixed respiratory and metabolic acidosis
✅ Answer: C. Acute-on-chronic respiratory acidosis
Reasoning: From his known baseline: PaCO₂ was 55 mmHg with appropriate chronic renal compensation. From baseline 55→72 = acute rise of 17 mmHg. For acute rise of 17 mmHg: Expected additional HCO₃⁻ = 0.1 × 17 = 1.7 → expected HCO₃⁻ = baseline + 1.7. His baseline HCO₃⁻ (from chronic compensation at PaCO₂ 55): expected = 24 + 0.35 × 15 = 29.25. So expected now = 29.25 + 1.7 ≈ 31. Measured = 34 → slightly above, but within range. The key is: this is ACUTE ON CHRONIC — the chronic component explains the high baseline HCO₃⁻, and the acute component explains the additional pH fall.
Q2: A 19-year-old woman presents with altered consciousness after ingesting an unknown substance. ABG: pH 7.22, PaCO₂ 18 mmHg, HCO₃⁻ 7 mEq/L. Na⁺ 140, Cl⁻ 107. Serum osmolality 340 mOsm/kg; calculated osmolality = 295 mOsm/kg. Which is the most likely toxin?
- A. Ethanol
- B. Methanol or ethylene glycol
- C. Isoniazid
- D. Salicylates
✅ Answer: B. Methanol or ethylene glycol
Reasoning: AG = 140 − (107 + 7) = 26 → HAGMA. Osmolal gap = 340 − 295 = 45 (>10) → elevated. Elevated osmolal gap + HAGMA = toxic alcohol until proven otherwise. Salicylates cause mixed respiratory alkalosis + metabolic acidosis and do not elevate osmolal gap significantly. Isoniazid causes lactic acidosis (HAGMA) but not osmolal gap elevation. Ethanol elevates osmolal gap but minimally contributes to AG. Distinguishing methanol vs ethylene glycol: methanol → formic acid, optic neuropathy, “snow blindness”; ethylene glycol → oxalic acid, renal failure, oxalate crystals in urine. Treatment: fomepizole (alcohol dehydrogenase inhibitor) + haemodialysis.
Q3: A 45-year-old with vomiting for 3 days has ABG: pH 7.52, PaCO₂ 48 mmHg, HCO₃⁻ 38 mEq/L. Urine Cl⁻ = 10 mEq/L. What does the urine chloride tell you and what is the treatment?
- A. Saline-resistant alkalosis; treat with spironolactone
- B. Saline-responsive alkalosis; treat with IV normal saline + KCl replacement
- C. Saline-resistant alkalosis; treat with acetazolamide
- D. Normal finding; no treatment for alkalosis needed
✅ Answer: B. Saline-responsive; IV normal saline + KCl
Reasoning: This is metabolic alkalosis (↑ pH, ↑ HCO₃⁻) from vomiting (HCl loss). Compensation: Expected PaCO₂ = 40 + 0.7 × 14 = 49.8 → measured 48 → appropriate compensation. Urine Cl⁻ = 10 mEq/L (<25) → saline-responsive (Cl⁻-avid state). In saline-responsive alkalosis: volume depletion → avid renal NaCl reabsorption → HCO₃⁻ co-reabsorbed to maintain electrochemical balance. Treatment: IV NaCl restores volume → kidneys can now excrete excess HCO₃⁻. KCl must be replaced concurrently (vomiting causes hypokalaemia which independently maintains alkalosis). If KCl not given, alkalosis persists despite saline.
Q4: A 35-year-old woman with recurrent renal stones has ABG: pH 7.30, PaCO₂ 30 mmHg, HCO₃⁻ 14 mEq/L. Na⁺ 140, Cl⁻ 118. Urine pH = 6.8. Serum K⁺ = 2.8 mEq/L. What is the diagnosis?
- A. Type 2 (Proximal) RTA — cannot reabsorb HCO₃⁻
- B. Type 1 (Distal) RTA — cannot secrete H⁺; urine pH persistently >5.5
- C. Type 4 (Hyperkalaemic) RTA — aldosterone deficiency
- D. Diarrhoea-induced NAGMA — high urine NH₄⁺
✅ Answer: B. Type 1 (Distal) RTA
Reasoning: NAGMA (AG = 140 − 118 − 14 = 8 → normal AG). Despite systemic acidaemia (pH 7.30), urine pH = 6.8 → cannot acidify urine below 5.5 → distal collecting duct cannot secrete H⁺ → Type 1 RTA. Hypokalaemia (K⁺ 2.8) → characteristic of Type 1 and Type 2 (both hypo, unlike Type 4 which is hyper). Recurrent renal stones (nephrocalcinosis) → because high urine pH precipitates CaHPO₄ and CaOx in alkaline urine. Type 2 RTA: urine pH CAN fall below 5.5 when serum HCO₃⁻ falls below the reabsorption threshold (~15 mEq/L), so urine pH would be acidic at this HCO₃⁻ level. Type 4: hyperkalaemia, not hypokalaemia.
Q5: A 32-year-old man is found unconscious at home. ABG: pH 7.18, PaCO₂ 52 mmHg, HCO₃⁻ 19 mEq/L. SpO₂ on pulse oximetry reads 98%. He has cherry-red skin. What is the diagnosis, and why is the SpO₂ misleading?
- A. Severe asthma exacerbation; SpO₂ unreliable due to bronchospasm
- B. Opioid overdose; SpO₂ unreliable due to vasodilation
- C. Carbon monoxide poisoning; SpO₂ unreliable because pulse oximetry cannot distinguish oxyhaemoglobin from carboxyhaemoglobin
- D. Methanol poisoning; SpO₂ unreliable due to formic acid interference
✅ Answer: C. Carbon monoxide poisoning; SpO₂ unreliable because CO-Hb reads as OxyHb
Reasoning: Cherry-red skin (CO-Hb gives cherry-red colour to blood and skin), mixed acidosis (elevated PaCO₂ from respiratory depression + elevated CO₂ from tissue hypoxia/lactic acidosis — HCO₃⁻ 19 lower than expected for pure respiratory acidosis → concurrent metabolic acidosis from tissue lactic acidosis). SpO₂ MISLEADING: Pulse oximetry uses two wavelengths of light that distinguish oxyhaemoglobin (OxyHb) from deoxyhaemoglobin. Carboxyhaemoglobin (COHb) absorbs light identically to OxyHb at 660 nm → standard pulse oximetry reads COHb as OxyHb → SpO₂ is falsely normal (98%) even with severe CO poisoning. ABG co-oximetry (measures 4+ Hb fractions directly) or a dedicated CO-oximeter (7-wavelength) can detect COHb. Treatment: 100% O₂ (reduces COHb t½ from 5h to 60–90 min) or hyperbaric O₂ (severe CO poisoning, neurological symptoms, pregnancy).
📚 References
📖 Ganong’s Review of Medical Physiology — Chapter 39: Acid-Base Balance
📖 Guyton and Hall Textbook of Medical Physiology — Chapters 30–31: Acid-Base Regulation
📖 Harrison’s Principles of Internal Medicine — Chapter 56: Acid-Base Disorders
📖 Marino’s The ICU Book — Section on Acid-Base Analysis
📖 Narins & Emmett, Medicine 1980 — Original compensation formula papers
📖 Davidson’s Principles and Practice of Medicine — Chapter on Fluid, Electrolyte, and Acid-Base Disorders
🚀 Keep Practising
ABG questions in NEET PG and USMLE require applying systematic reasoning under time pressure. Practise the 5-step method on every ABG you see in clinical rotations. The fastest route to mastery: know the compensation formulae cold, always calculate the anion gap when HCO₃⁻ is low, and always check if compensation is appropriate.