What You Will Learn in This Article
- Define tinnitus precisely and classify it into all major subtypes
- Explain the neurophysiological and cochlear mechanisms underlying tinnitus generation
- List every major cause of tinnitus — otic, neurological, vascular, metabolic, and drug-related
- Differentiate pulsatile from non-pulsatile tinnitus and identify the red-flag features that demand urgent investigation
- Describe the audiological investigations used to evaluate tinnitus
- Apply the Jastreboff neurophysiological model and understand how it guides treatment
- Outline all current treatment strategies: TRT, CBT, sound therapy, pharmacology, and surgical options
- Connect tinnitus to the major ENT conditions it accompanies — Ménière disease, acoustic neuroma, otosclerosis, NIHL
📖 Introduction: Why This Topic Matters in Exams
A 52-year-old construction worker presents with a persistent ringing sound in both ears that has been present for 2 years. He reports he has difficulty hearing conversations in noisy places and that the ringing is worse after a full day at work. His audiogram shows a characteristic notch at 4000 Hz. He has no vertigo, no facial weakness, and no pulsatile quality to the sound. This is noise-induced hearing loss with associated tinnitus — one of the most common and most examinable presentations in ENT. Understanding tinnitus means understanding a window into the entire auditory system.
Tinnitus — the perception of sound without an external acoustic stimulus — affects approximately 10–15% of the adult population and is one of the most common reasons patients consult an ENT surgeon. In medical entrance exams, tinnitus questions appear as part of clinical scenarios on Ménière disease, acoustic neuroma, otosclerosis, drug ototoxicity, and noise-induced hearing loss. They also appear as direct questions on the audiological basis of tinnitus and on its management. Understanding tinnitus requires integrating anatomy, physiology, pharmacology, and clinical medicine — which is exactly why it is a favourite exam topic.
This article covers all essential aspects: definition, classification, pathophysiology, causes, red flags, investigations, and the full management spectrum. By the end, you will be able to handle any tinnitus question in both theoretical and clinical formats.
🔬 Section 1 — Definition and Classification
Definition
Tinnitus (Latin: tinnire — to ring) is defined as the conscious perception of a sound in the absence of an external acoustic stimulus. It is a symptom, not a disease — it reflects abnormal neural activity somewhere in the auditory pathway.
The sound can be described as:
- Ringing (most common)
- Buzzing, humming, hissing
- Clicking, pulsing, roaring
- Musical (rare) — “musical hallucinations”
Classification
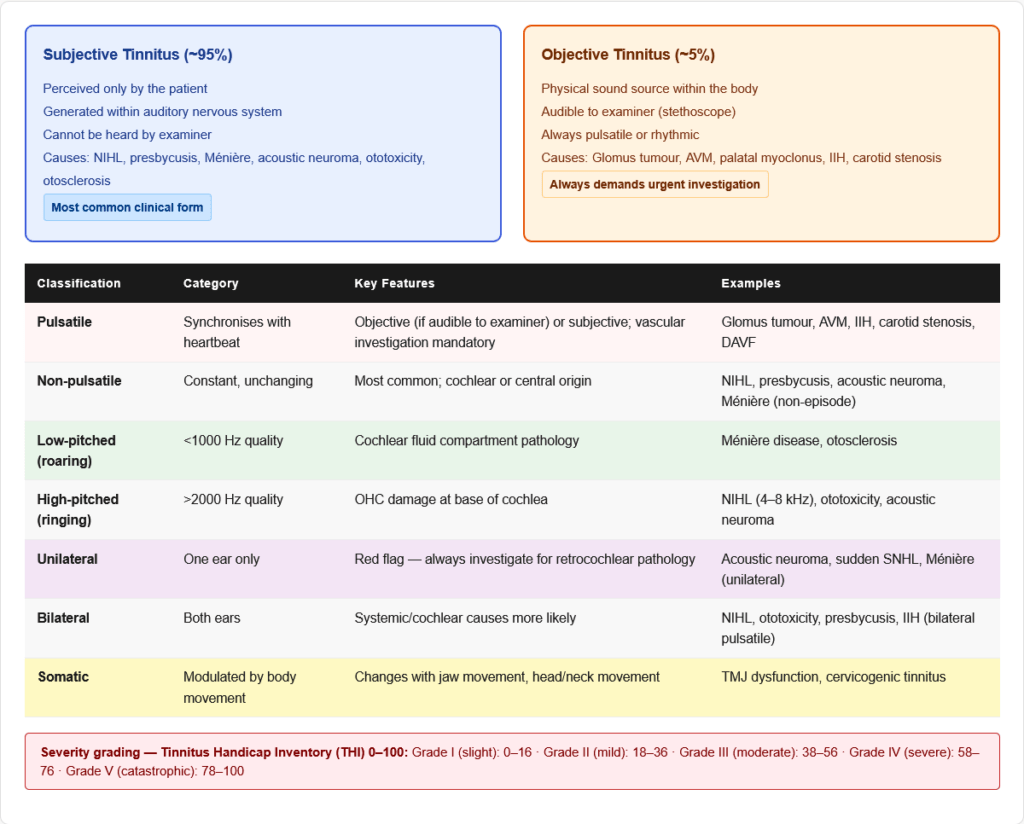
1. By Audibility: Subjective vs Objective
| Type | Definition | Audible to Examiner? | Prevalence |
|---|---|---|---|
| Subjective Tinnitus | Perceived only by the patient — generated within the auditory nervous system | No | ~95% |
| Objective Tinnitus | Generated by a physical sound source within the body — can be heard by the examiner with a stethoscope | Yes | ~5% |
Objective tinnitus causes (important — always pulsatile or rhythmic):
- Vascular: Arteriovenous malformations, dural arteriovenous fistula, carotid stenosis, glomus tumour (paraganglioma), venous hum from jugular bulb
- Mechanical: Palatal myoclonus (rhythmic clicking — audible to examiner), tensor tympani myoclonus, stapedial myoclonus, patulous Eustachian tube
- Intracranial hypertension (IIH) — venous pulsatile tinnitus
2. By Quality: Pulsatile vs Non-Pulsatile
Pulsatile tinnitus — synchronises with the heartbeat; always demands vascular investigation
| Pulsatile Tinnitus Cause | Key Feature |
|---|---|
| Glomus jugulare/tympanicum (paraganglioma) | Red mass behind tympanic membrane; pulsatile; biopsy = extreme danger (haemorrhage) |
| Arteriovenous malformation (AVM) | Objective — audible to examiner; MRI/MRA for diagnosis |
| Carotid artery stenosis / ICA aneurysm | Unilateral; risk of stroke; Doppler/CTA |
| Dural arteriovenous fistula (DAVF) | Whooshing, unilateral; MRA/conventional angiography |
| Idiopathic Intracranial Hypertension (IIH) | Young obese women; bilateral pulsatile tinnitus + papilloedema; normal MRI then LP (elevated opening pressure) |
| High jugular bulb / Dehiscent jugular bulb | Pulsatile; visible bluish mass in hypotympanum |
| Carotid-cavernous fistula | Pulsatile proptosis + chemosis + orbital bruit |
| Venous hum | Disappears with ipsilateral jugular vein compression |
| Benign causes | Anaemia (high-flow state), pregnancy, hyperthyroidism |
Non-pulsatile tinnitus — the vast majority; caused by cochlear or central auditory pathway pathology
3. By Duration
- Acute tinnitus: <3 months
- Subacute tinnitus: 3–12 months
- Chronic tinnitus: >12 months
4. By Distribution
- Unilateral: Suggests localised cochlear disease or acoustic neuroma — ALWAYS investigate
- Bilateral: Suggests systemic causes (NIHL, ototoxicity, Ménière, presbycusis)
- Central (inside the head): Suggests central auditory pathway involvement
5. By Severity
Tinnitus Handicap Inventory (THI): A 25-item validated questionnaire measuring functional, emotional, and catastrophic impact. Scores 0–100:
- Grade I (slight): 0–16
- Grade II (mild): 18–36
- Grade III (moderate): 38–56
- Grade IV (severe): 58–76
- Grade V (catastrophic): 78–100
🔬 Section 2 — Anatomy of the Auditory Pathway

Understanding where tinnitus arises requires a clear map of the auditory system:
Peripheral Auditory System
Outer ear → Middle ear → Cochlea (Inner ear):
- Sound waves → external auditory canal → tympanic membrane → ossicular chain (malleus → incus → stapes)
- Stapes footplate → oval window → cochlear fluid waves (scala vestibuli → scala media → scala tympani)
- Basilar membrane vibration — tonotopic organisation: High frequencies near the base; Low frequencies near the apex
- Hair cells (inner and outer) in the organ of Corti:
- Inner hair cells (IHC): ~3,500; the primary sensory transducers — convert mechanical vibration to electrical signals; each IHC connects to ~10–20 afferent nerve fibres
- Outer hair cells (OHC): ~12,000; electromotile — amplify basilar membrane motion (cochlear amplifier); key to frequency selectivity and audiogram “notch” in NIHL (OHC damaged first at the 4 kHz region)
- Mechanotransduction: Stereocilia deflection → opens tip-link gated ion channels → K⁺/Ca²⁺ influx → depolarisation → neurotransmitter (glutamate) release → action potential in cochlear nerve (CN VIII)
Central Auditory Pathway
Cochlear nerve (CN VIII) → Cochlear nucleus (brainstem) → Superior olivary complex (binaural processing) → Inferior colliculus (midbrain) → Medial geniculate nucleus (thalamus) → Primary auditory cortex (Heschl gyrus, temporal lobe)
The limbic system and prefrontal cortex have important connections to the auditory cortex — this explains the emotional and psychological dimensions of tinnitus.
⚙️ Section 3 — Pathophysiology of Tinnitus
Why Does Tinnitus Occur? — Key Mechanisms
Tinnitus is fundamentally a problem of neural activity without corresponding sound input — aberrant signals at some level of the auditory pathway. Multiple mechanisms operate simultaneously:
1. Loss of Peripheral Inhibition (Cochlear Deafferentation Model)
The most fundamental and best-supported mechanism:
- Normally, the auditory cortex receives a constant pattern of spontaneous neural activity from the cochlea — this acts as a “reference baseline”
- When cochlear hair cells are damaged (NIHL, ototoxicity, aging), afferent input from that frequency region is lost or reduced
- The central auditory neurons that previously received input from the damaged region become hyperactive — they fire spontaneously at high rates in an attempt to compensate for the lost input (analogous to phantom limb pain after amputation)
- This central gain increase generates the perception of sound (tinnitus) even though the cochlea is no longer sending signals at that frequency
- This is why tinnitus frequency often matches the audiometric notch — the cortical region deprived of input generates its own “signal”
- Key analogy: When a power outage occurs and one circuit loses power, the neighbouring circuits may generate background noise — this is what the brain does when cochlear input is lost
2. Maladaptive Neuroplasticity and Cortical Reorganisation
- After cochlear damage, the brain tries to reorganise:
- Neurons in the deprived frequency area begin responding to adjacent frequencies
- This tonotopic map reorganisation creates aberrant cross-frequency connections
- The reorganised cortex is unstable → generates spurious activity → tinnitus
- This is similar to the cortical map changes seen in phantom limb pain and chronic pain
3. Increased Spontaneous Activity in Cochlear Nucleus and IC
- Dorsal cochlear nucleus (DCN) neurons show increased spontaneous discharge rates after cochlear damage
- The inferior colliculus (IC) — a key brainstem auditory station — shows hyperactivity correlated with tinnitus
- This hyperactivity propagates up the pathway to auditory cortex
4. Dysfunction of the Descending (Efferent) System
- The medial olivocochlear bundle (MOCB) normally provides inhibitory feedback to outer hair cells — it is a “volume control” for cochlear amplification
- Damage to OHCs or the efferent system → loss of this inhibitory feedback → sustained cochlear amplification → tinnitus
- MOCB also suppresses contralateral cochlear responses — its dysfunction may contribute to tinnitus asymmetry
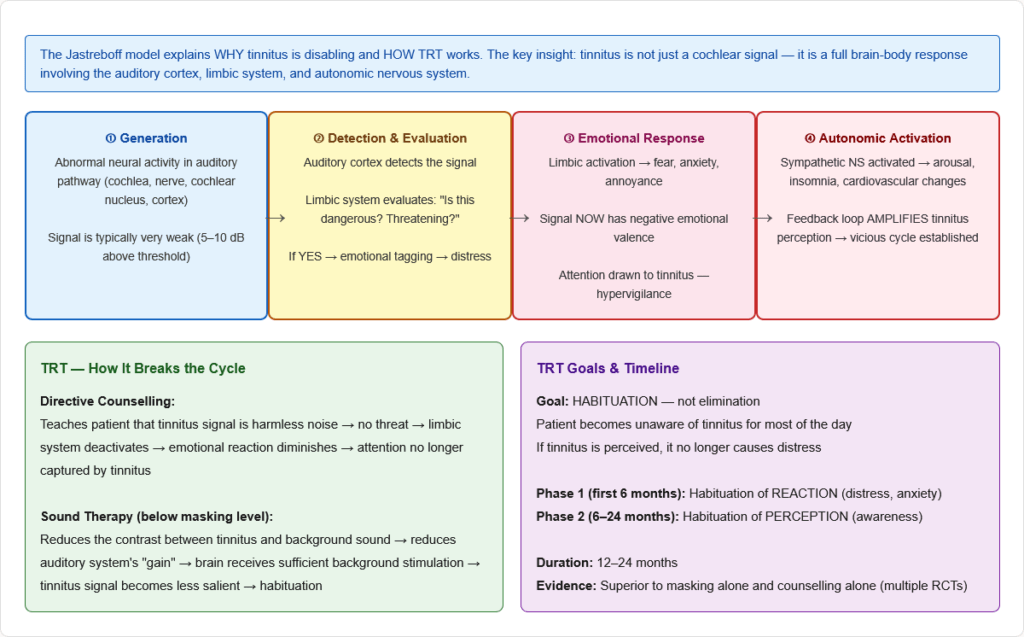
5. The Jastreboff Neurophysiological Model
Jastreboff (1990) proposed that tinnitus is NOT just a cochlear or auditory cortex problem — it involves the entire auditory-limbic-autonomic nervous system network:
- Generation: Abnormal neural activity (from any auditory level)
- Detection: Auditory cortex detects the signal
- Emotional evaluation: Limbic system assigns negative emotional significance (fear, anxiety, annoyance)
- Autonomic activation: Sympathetic activation → arousal, hypervigilance, stress response
- Feedback loop: Emotional distress and autonomic activation AMPLIFY the perception of tinnitus → “conditioned reflex arc” is established
- The tinnitus is not the problem — the REACTION to the tinnitus is the clinical problem
- This model is the basis of Tinnitus Retraining Therapy (TRT)
6. Cochlear Origin Mechanisms
In some cases, tinnitus arises from the cochlea itself:
- Spontaneous otoacoustic emissions (SOAEs): Occasional acoustic energy spontaneously generated by OHCs can be amplified to conscious perception
- Hair cell damage: Partially damaged hair cells depolarise spontaneously → drive auditory nerve → tinnitus
- Changes in endolymph: Altered endolymphatic pressure (Ménière disease) → changes hair cell mechanotransduction → tinnitus
- Synaptic dysfunction: Cochlear synaptopathy (damage to IHC-nerve synapses without hair cell loss) — “hidden hearing loss” — now recognised as an important cause, especially from noise and aging
🏥 Section 4 — Causes of Tinnitus
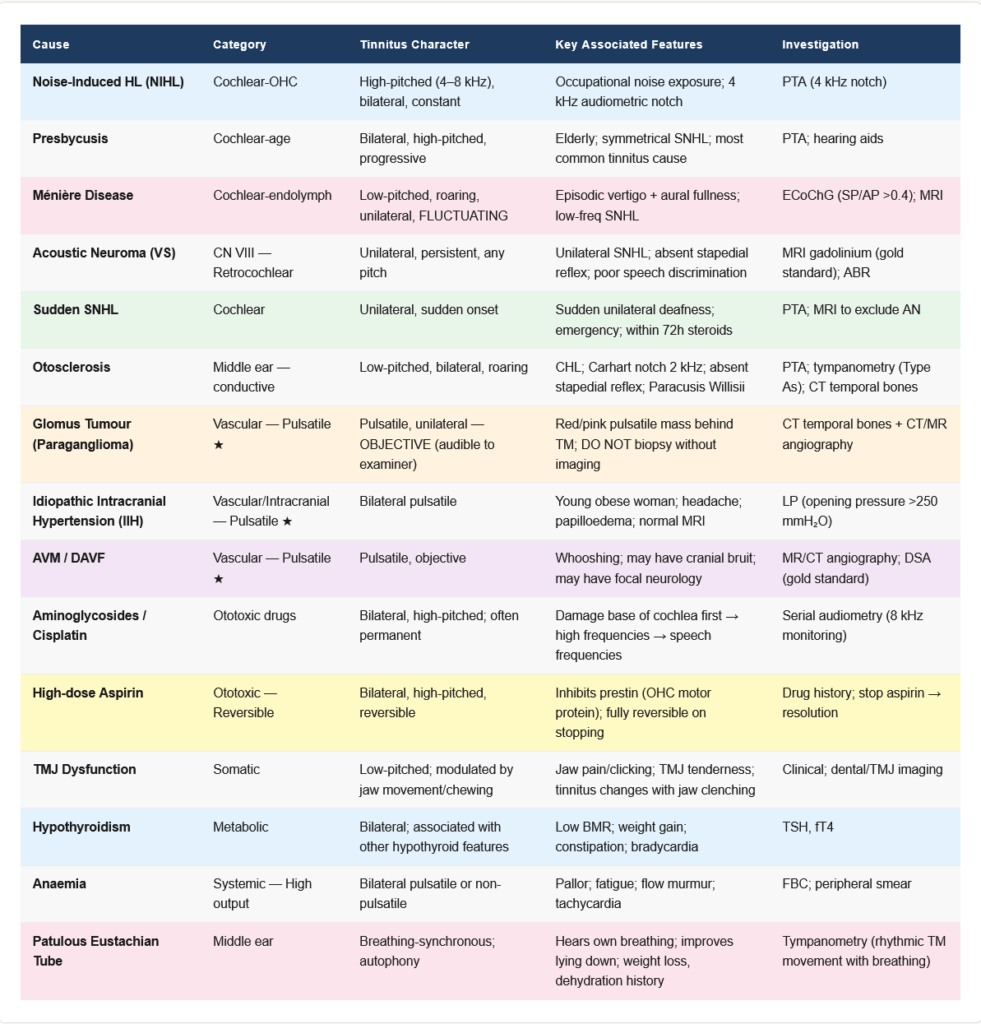
1. Otic (Ear) Causes — The Most Common Category
Sensorineural causes:
| Condition | Tinnitus Character | Associated Features |
|---|---|---|
| Noise-Induced Hearing Loss (NIHL) | High-pitched, bilateral, constant | 4 kHz notch on audiogram; occupational/recreational noise |
| Presbycusis (Age-related HL) | Bilateral, high-pitched | Gradual onset; symmetrical SNHL; elderly |
| Ménière Disease | Low-pitched, fluctuating, unilateral | Triad: episodic vertigo + tinnitus + SNHL; aural fullness |
| Acoustic Neuroma (Vestibular Schwannoma) | Unilateral, persistent | Unilateral SNHL, unilateral tinnitus, poor speech discrimination |
| Sudden SNHL | Unilateral, sudden onset | Emergency — IV steroids within 72 hours |
| Ototoxicity | Bilateral, high-pitched | Drug history (aminoglycosides, cisplatin, loop diuretics) |
| Cochlear otosclerosis | Tinnitus + low-frequency HL | Pagetoid changes in cochlear capsule |
Conductive causes:
| Condition | Mechanism | Tinnitus Character |
|---|---|---|
| Otosclerosis | Stapedial fixation → CHL + SNHL component | Low-pitched, bilateral, roaring |
| Otitis media (chronic) | Middle ear pathology | Varies; often low-pitched |
| Cerumen impaction | Occlusion of EAC | Reversible; resolves with removal |
| Eustachian tube dysfunction | Middle ear pressure changes | Fluctuating |
| Patulous ET | Open ET → autophony, respiratory tinnitus | Breathing-synchronous tinnitus; improves lying down |
| Tympanosclerosis | Calcification of middle ear | Associated CHL |
2. Neurological Causes
- Acoustic neuroma (VS) — the most feared unilateral tinnitus cause; 8th nerve; MRI with gadolinium is gold standard
- Multiple sclerosis — demyelination of auditory pathway
- Meningitis — can damage cochlea
- Head trauma — cochlear concussion, labyrinthine haemorrhage
- Intracranial tumours — CPA angle tumours, meningioma
- Migraine-associated auditory disturbance — tinnitus as migraine aura
3. Vascular Causes (Pulsatile Tinnitus)
As listed above — all pulsatile tinnitus demands vascular workup.
4. Metabolic Causes
| Condition | Mechanism | Test |
|---|---|---|
| Anaemia | High cardiac output → increased turbulence in vessels near cochlea | FBC |
| Hypothyroidism | Altered cochlear metabolism; endolymphatic hydrops-like changes | TSH, T4 |
| Hyperthyroidism | Increased cardiac output; peripheral vasodilatation | TSH |
| Diabetes mellitus | Diabetic angiopathy of cochlear vessels; neuropathy | HbA1c |
| Hyperlipidaemia | Sludging of cochlear microcirculation | Lipid profile |
| Vitamin B12 deficiency | Auditory neuropathy | B12 levels |
| Zinc deficiency | Required for cochlear enzyme function | Zinc levels |
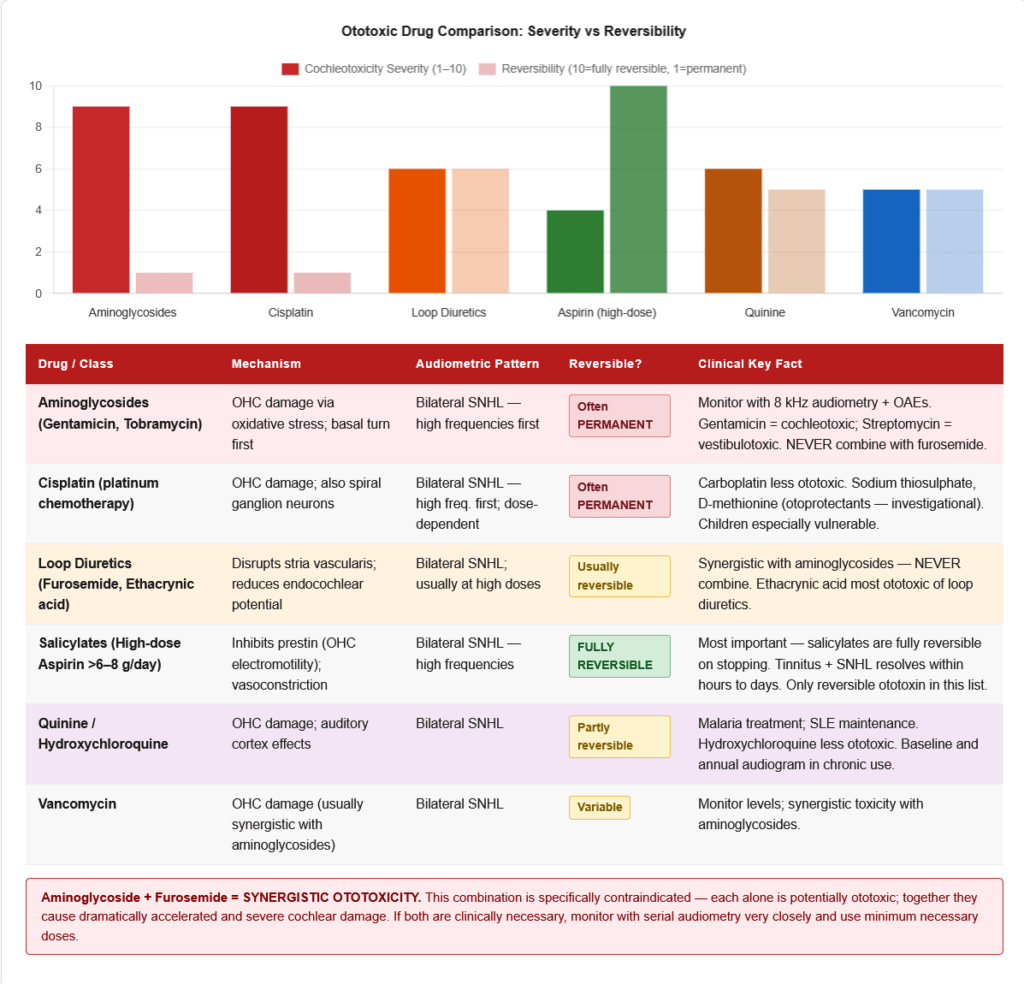
5. Drug-Induced Tinnitus (Ototoxicity)
This is a high-yield area — multiple drug classes cause tinnitus and hearing loss:
| Drug Class | Examples | Mechanism | Reversibility |
|---|---|---|---|
| Aminoglycosides | Gentamicin, tobramycin, neomycin | OHC damage (primarily cochleotoxic for gentamicin; vestibulotoxic for streptomycin) | Often permanent |
| Cisplatin | Cisplatin (carboplatin less so) | OHC damage — basal turn first → high-frequency loss | Often permanent; dose-dependent |
| Loop diuretics | Furosemide, ethacrynic acid | Disrupts endocochlear potential (stria vascularis); reversible at normal doses; permanent at high doses | Usually reversible |
| Salicylates (high dose) | Aspirin >6–8 g/day | Reversible inhibition of OHC electromotility (prestin); also affects blood flow | Fully reversible on stopping |
| Quinine / antimalarials | Quinine, chloroquine, hydroxychloroquine | OHC damage; auditory cortex effects | Partially reversible |
| NSAIDs | Ibuprofen, naproxen | Mild OHC effects; usually reversible | Reversible |
| Vancomycin | Vancomycin | Synergistic toxicity with aminoglycosides | Variable |
| Antidepressants | TCAs, SSRIs (less common) | Central auditory pathway effects | Usually reversible |
| Methotrexate | High-dose | Cochlear toxicity | Variable |
Combined ototoxicity: Aminoglycosides + loop diuretics is particularly dangerous — synergistic cochleotoxicity. Never co-prescribe without careful monitoring.
6. Temporomandibular Joint (TMJ) Dysfunction
- The TMJ shares anatomical proximity to the middle ear and Eustachian tube orifice
- TMJ dysfunction → referred tinnitus; usually low-pitched, associated with jaw pain/clicking, worse with chewing
- This is a somatic tinnitus — modulated by jaw movement (a distinguishing clinical feature)
7. Cervicogenic Tinnitus
- Tinnitus associated with cervical spine pathology or muscle dysfunction
- Modulated by head and neck movements
- Probably mediated via somatosensory input to the cochlear nucleus (the DCN receives significant somatosensory input from the trigeminal and dorsal horn neurons)
8. Psychological Associations
- Tinnitus has very high rates of comorbidity with anxiety, depression, and PTSD
- The relationship is bidirectional: tinnitus causes psychological distress, and psychological distress amplifies tinnitus perception
- Tinnitus severity as measured by THI correlates more strongly with psychological distress than with audiological parameters
🔬 Section 5 — Red Flag Features: When Tinnitus is an Emergency

The following features in a patient with tinnitus demand urgent investigation:
Red flags requiring urgent MRI/investigation:
- Unilateral tinnitus — especially without obvious cause (exclude acoustic neuroma)
- Pulsatile tinnitus — exclude vascular cause; CT/MR angiography
- Tinnitus with rapid progressive hearing loss — sudden SNHL or acoustic neuroma
- Tinnitus with vertigo — Ménière disease, acoustic neuroma, labyrinthitis
- Tinnitus with neurological signs — facial nerve palsy, ataxia, headache → posterior fossa tumour
- Tinnitus with papilloedema — Idiopathic Intracranial Hypertension (IIH)
- Pulsatile tinnitus with red mass behind eardrum — Glomus tumour (do NOT biopsy blindly)
- Tinnitus in a child — rarely physiological; always investigate
🔬 Section 6 — Investigations for Tinnitus
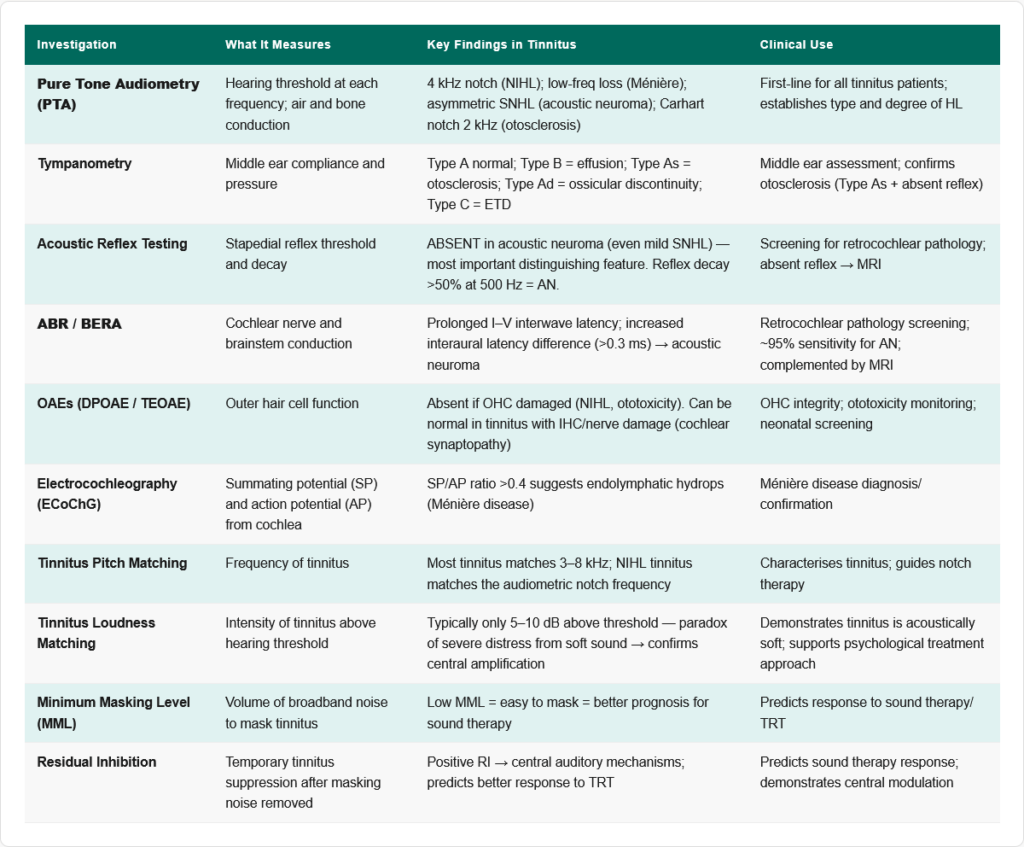
Audiological Assessment
1. Pure Tone Audiometry (PTA)
- Establishes type and degree of hearing loss (conductive vs sensorineural)
- The audiogram pattern guides differential diagnosis:
- 4 kHz notch → NIHL
- Flat SNHL with low-frequency component → Ménière disease
- Asymmetric SNHL → acoustic neuroma
- Carhart notch (2 kHz dip) → otosclerosis
- Air-bone gap: Conductive HL has air-bone gap >10 dB; SNHL has no air-bone gap
2. Tympanometry
- Type A (normal): Peak compliance at 0 daPa — normal middle ear
- Type B (flat): No peak — middle ear effusion or perforation
- Type C (negative pressure): ETD
- Type Ad (hypercompliant): Ossicular discontinuity, or flaccid TM
- Type As (reduced compliance): Otosclerosis, tympanosclerosis
3. Acoustic Reflex Testing
- Stapedial reflex absent in acoustic neuroma (even with mild SNHL) — important diagnostic pointer
- Absent in facial nerve palsy (proximal to stapedius branch of CN VII)
- Stapedial reflex decay test: >50% decay at 500 Hz or 1000 Hz → acoustic neuroma
4. Otoacoustic Emissions (OAEs)
- DPOAEs (Distortion Product OAEs): Measure OHC function; absent if OHC damaged
- TEOAEs (Transient Evoked OAEs): Present in normal hearing; absent in SNHL >30 dB
- Spontaneous OAEs (SOAEs): Occasionally measurable in some objective tinnitus cases
- OAEs can be normal even when tinnitus is present — they test OHC, not IHC or nerve
5. Auditory Brainstem Response (ABR / BERA)
- Tests integrity of cochlear nerve and brainstem auditory pathway
- Most important test for acoustic neuroma detection:
- Prolonged Wave I–III and I–V interpeak latencies
- Interaural latency difference >0.3 ms
- Absent or abnormal waveforms
- Sensitivity ~95% for acoustic neuroma; complemented by MRI
6. Electrocochleography (ECoChG)
- Records cochlear receptor potentials (SP — summating potential; AP — action potential)
- SP/AP ratio >0.4 suggests endolymphatic hydrops (Ménière disease)
7. Tinnitus Pitch and Loudness Matching
- Ask patient to match the pitch of their tinnitus to external tones
- Most tinnitus is in the 3–8 kHz range (high-pitched)
- Loudness matching: tinnitus is typically only 5–10 dB above hearing threshold — very soft sounds, yet highly intrusive → reflects central amplification
8. Minimum Masking Level (MML)
- The minimum intensity of broadband noise needed to mask tinnitus
- Low MML → easy to mask → better prognosis for sound therapy
9. Residual Inhibition
- After masking noise is turned off, tinnitus is temporarily reduced or absent
- Positive residual inhibition (RI) → indicates central auditory mechanisms involved; predicts response to sound therapy/TRT
Imaging
| Investigation | When to Use | What It Shows |
|---|---|---|
| MRI with gadolinium (brain + internal auditory canals) | Unilateral tinnitus, asymmetric SNHL, suspected acoustic neuroma | Gold standard for acoustic neuroma; CPA angle lesions |
| CT temporal bones | Suspected otosclerosis, cholesteatoma, glomus tumour, temporal bone fracture | Bony detail of middle ear, cochlear capsule, jugular bulb |
| MR angiography (MRA) | Pulsatile tinnitus | AVM, DAVF, carotid artery disease |
| CT angiography (CTA) | Urgent pulsatile tinnitus, suspected AVM | Vascular malformations |
| Carotid Doppler | Pulsatile tinnitus, cardiovascular risk factors | Carotid stenosis |
| Cerebral angiography (DSA) | Gold standard for AVM/DAVF if MRA inconclusive | Definitive vascular mapping |
Laboratory Tests
- FBC (anaemia), TFTs (thyroid disease), fasting glucose/HbA1c (diabetes), lipid profile, B12 and folate, zinc levels, renal function (if ototoxic drugs given), syphilis serology (FTA-ABS — syphilitic labyrinthitis is treatable)
- Autoimmune panel if suspected autoimmune inner ear disease (AIED): ANA, anti-dsDNA, anti-cochlear antibodies
⚙️ Section 7 — Management of Tinnitus
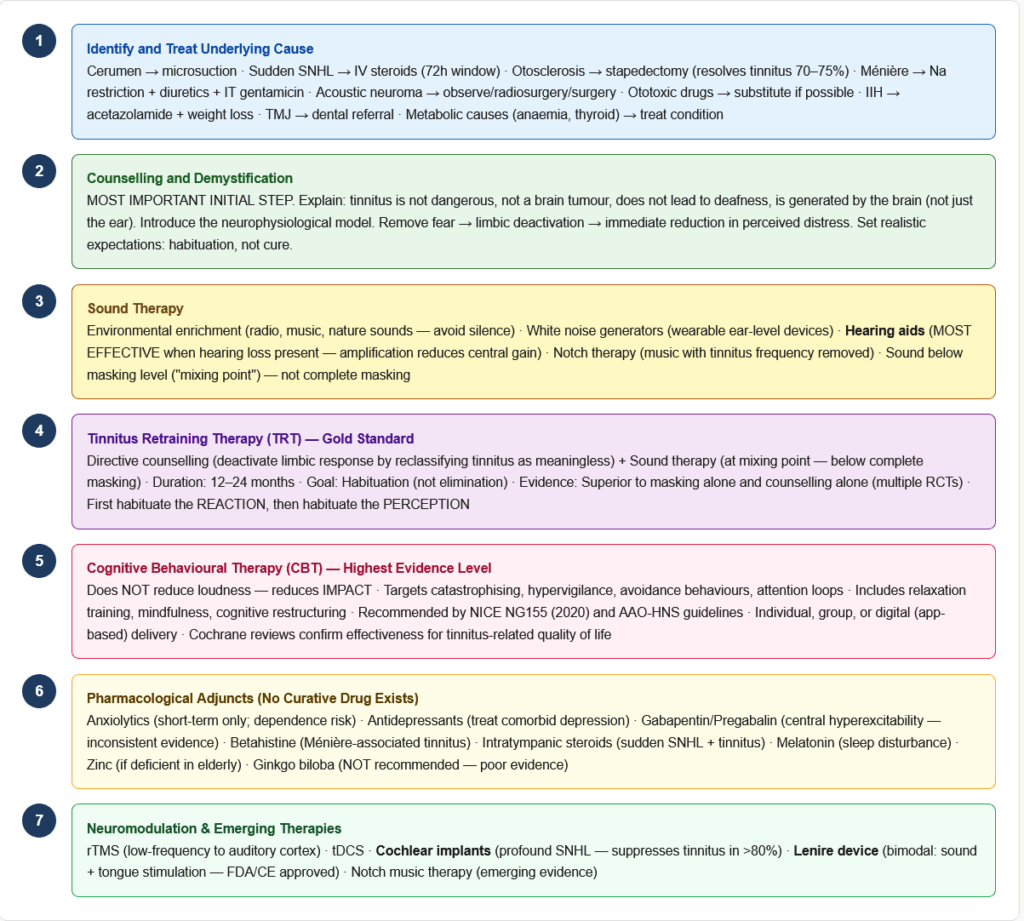
The Fundamental Principle
There is no universally effective pharmacological cure for tinnitus. Management focuses on:
- Identifying and treating the underlying cause (when possible)
- Reducing the impact of tinnitus on quality of life
- Using sound therapy to reduce auditory system hyperactivity
- Addressing psychological components (anxiety, depression)
Step 1: Treat the Underlying Cause
- NIHL → hearing protection; workplace audiometry; no cure for existing damage
- Cerumen → microsuction/syringe → immediate resolution of tinnitus in some
- Acute otitis media → antibiotics
- Sudden SNHL → systemic corticosteroids urgently (within 72 hours)
- Otosclerosis → stapedectomy → resolves tinnitus in 70–75% of cases
- Acoustic neuroma → observation (small), radiosurgery (Gamma Knife), or surgery
- Ménière disease → dietary sodium restriction, diuretics, intratympanic steroids/gentamicin
- Ototoxic drugs → substitute or reduce where clinically possible
- TMJ dysfunction → dental/physiotherapy, splinting
- Thyroid disorders, anaemia → treat underlying metabolic cause
- IIH → acetazolamide, weight loss; LP for diagnosis and temporary relief
Step 2: Counselling and Demystification
The most important initial intervention — many patients fear tinnitus indicates a brain tumour, impending deafness, or madness:
- Thorough explanation of what tinnitus is and why it occurs
- Reassurance that tinnitus is not dangerous (when cause is benign) and does not indicate progressive neurological disease
- Education about the neurophysiological model — understanding removes fear → reduces limbic system activation → reduces tinnitus distress
- Setting realistic expectations: habituation is the goal, not elimination
Step 3: Sound Therapy
Rationale: Reducing the contrast between tinnitus and background sound reduces the relative salience of tinnitus to the brain.
Types of sound therapy:
- Environmental sound enrichment: Keeping the environment acoustically rich (radio, music, nature sounds) — most basic intervention
- White noise generators / Sound maskers: Wearable ear-level devices that emit broadband noise to partially mask tinnitus
- Hearing aids: In patients with hearing loss — amplifying ambient sound reduces auditory deprivation and reduces central gain; most effective tool when HL is present
- Notch therapy: Music with the tinnitus frequency removed (notched) — reduces cortical synchrony at the tinnitus frequency; emerging evidence
- Neuromodulation via sound: Specifically designed protocols to target cortical reorganisation
Step 4: Tinnitus Retraining Therapy (TRT)
The gold standard evidence-based treatment — developed by Jastreboff and Hazell based on the neurophysiological model:
Two components:
- Directive counselling — teaching the patient that tinnitus signals are meaningless background noise, not a threat; re-framing the limbic response from negative to neutral
- Sound therapy — constant low-level broadband sound (below the masking level — “mixing point”) to reduce auditory contrast, reduce central gain, and facilitate habituation
Goal: Habituation — the patient becomes unaware of tinnitus most of the time, similar to how we habituate to other meaningless stimuli (a clock ticking, clothing pressure)
Duration: 12–24 months; results maintained long-term in most patients
Evidence: Multiple RCTs show TRT superior to masking alone and to counselling alone. It is effective regardless of tinnitus severity.
Step 5: Cognitive Behavioural Therapy (CBT)
Highest level of evidence for improving tinnitus-related quality of life:
- Does not reduce the loudness of tinnitus
- Reduces the emotional and psychological impact
- Targets catastrophising thoughts, avoidance behaviours, hypervigilance
- Includes relaxation training, attention redirection, cognitive restructuring
- Delivered individually or in groups; increasingly available in digital (app-based) formats
- NICE (UK) and AAO-HNS guidelines recommend CBT as a core treatment modality
Step 6: Pharmacological Approaches
No drug has robust level I evidence for tinnitus treatment. However:
| Drug | Status | Evidence | Notes |
|---|---|---|---|
| Anxiolytics / Benzodiazepines | Symptomatic only | Short-term relief of distress; not for long-term use | Dependence risk; clonazepam sometimes used |
| Antidepressants (TCAs, SSRIs) | Treat comorbid depression | No direct anti-tinnitus effect; but depression worsens tinnitus | Amitriptyline — oldest studied |
| Gabapentin / Pregabalin | Some studies positive | Reduces central neural hyperexcitability | Inconsistent results; may help in specific types |
| Betahistine | Used in Ménière-associated tinnitus | Limited evidence for tinnitus per se; helps vertigo and hearing in Ménière | Histamine H1 agonist / H3 antagonist |
| Intratympanic steroids | For sudden SNHL + tinnitus | Good evidence for SNHL; variable for tinnitus | Dexamethasone IT |
| Zinc supplementation | Zinc-deficient elderly patients | Some benefit | Only if deficient |
| Ginkgo biloba | Widely used; poor evidence | Not recommended by most guidelines | Inconsistent studies |
| Melatonin | Helps with sleep disruption from tinnitus | Limited direct anti-tinnitus effect | Useful adjunct if insomnia is prominent |
| Lidocaine IV | Temporarily suppresses tinnitus in some | Not practical for long-term use | Used in research to confirm central mechanisms |
Step 7: Neuromodulation and Emerging Therapies
Transcranial Magnetic Stimulation (rTMS):
- Low-frequency rTMS to auditory cortex → reduces cortical hyperactivity
- Moderate evidence; effect wanes after treatment ends
- Not widely available; used in specialist centres
Transcranial Direct Current Stimulation (tDCS):
- Non-invasive cortical modulation
- Some evidence for reducing tinnitus loudness
Deep Brain Stimulation (DBS):
- Investigational; targeting auditory thalamus
- Used only in refractory severe cases
Cochlear Implants:
- For patients with severe-profound SNHL + severe tinnitus
- Strong evidence: cochlear implantation suppresses tinnitus in the vast majority of suitable patients
- The electrical stimulation from the implant provides auditory input that suppresses central hyperactivity
Bimodal Stimulation (Lenire device):
- Combines sound stimulation with tongue electrical stimulation (cranial nerve stimulation)
- FDA/CE approved (first approved device specifically for tinnitus)
- Based on paired associative stimulation to drive cortical plasticity
Step 8: Lifestyle and Self-Management
- Hearing protection: Essential in noise-exposed individuals
- Caffeine and alcohol: Evidence is mixed; some patients report worsening; advise moderation
- Exercise: General health benefits; some evidence that cardiovascular exercise reduces tinnitus severity
- Sleep hygiene: Tinnitus is worst in silence at night; sound therapy/white noise machine at bedside is helpful
- Mindfulness-based stress reduction (MBSR): Evidence for improving quality of life with tinnitus
- Support groups: Peer support; normalisation; British Tinnitus Association (BTA) / American Tinnitus Association (ATA)
🏥 Section 8 — Tinnitus in Specific Clinical Conditions
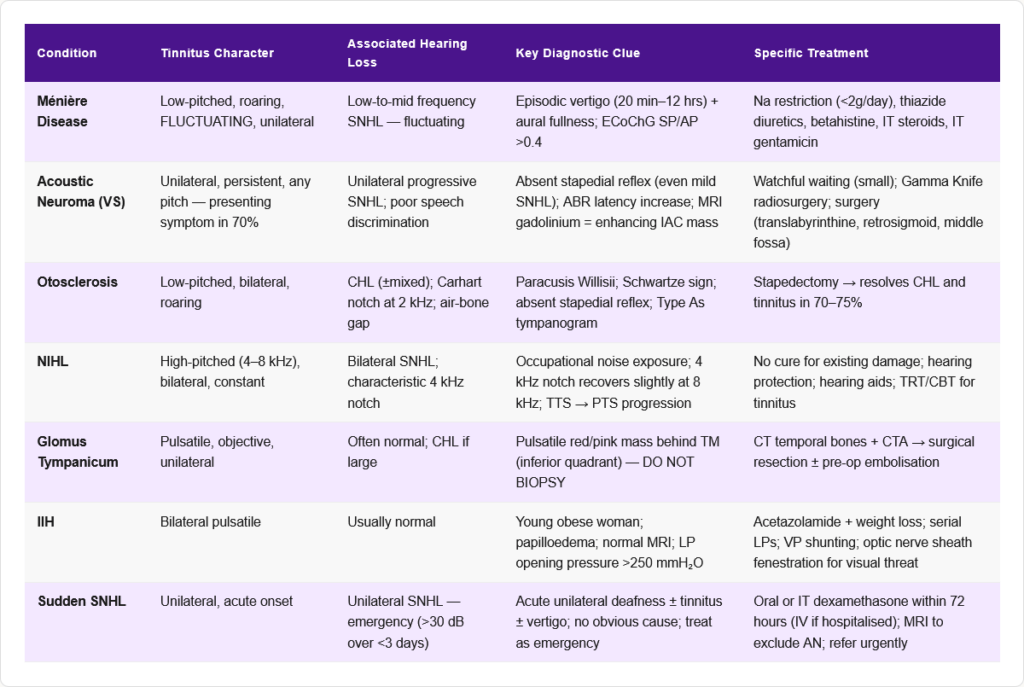
Ménière Disease
- Tinnitus in Ménière is classically low-pitched, roaring, fluctuating, and unilateral
- Fluctuates with episodes — often louder before and during a vertiginous attack
- The underlying cause is endolymphatic hydrops (excessive endolymph → distension of membranous labyrinth → altered hair cell mechanotransduction)
- Diagnostic criteria (2015 AAO-HNS): ≥2 definitive episodes of vertigo lasting 20 min–12 hours, documented low-to-mid frequency SNHL on audiogram, fluctuating aural symptoms (tinnitus, fullness, hearing) in the affected ear, not attributed to another diagnosis
- Treatment of Ménière tinnitus: dietary sodium restriction (<2g/day), thiazide diuretics, betahistine, intratympanic gentamicin (chemical labyrinthectomy for refractory vertigo)
Acoustic Neuroma (Vestibular Schwannoma)
- Unilateral tinnitus is the presenting symptom in 70% of acoustic neuromas
- The tumour arises from the Schwann cells of the vestibular (not acoustic) division of CN VIII within the internal auditory canal
- Other features: progressive unilateral SNHL (>95%), absent stapedial reflex, cerebellar signs if large
- Investigation: MRI with gadolinium (gold standard) — shows enhancing mass in IAC/CPA; ABR shows interaural latency difference
- Treatment: Watchful waiting (small tumours in elderly), stereotactic radiosurgery (Gamma Knife), or surgical resection (translabyrinthine, retrosigmoid, or middle fossa approach)
Otosclerosis
- Tinnitus in otosclerosis is typically low-pitched, bilateral (often), roaring
- Associated with conductive hearing loss (±mixed) — Carhart notch at 2 kHz on audiogram
- On tympanometry: Type As (reduced compliance — stiff TM due to fixed stapes)
- Stapedial reflex is absent (fixed stapes cannot contract stapedius)
- Paracusis Willisii (hearing better in noise — because normal-hearing people raise their voices in noise, levelling the playing field) — characteristic of otosclerosis
- Schwartze sign: Flamingo-pink blush seen through TM — active otospongiosis with vascular bone
- Treatment: Stapedectomy → replaces fixed stapes with prosthesis → resolves CHL and significantly reduces tinnitus in ~70–75%
Noise-Induced Hearing Loss (NIHL)
- Most common preventable cause of SNHL worldwide
- Mechanism: Sound energy at high intensities (>85 dB SPL) damages outer hair cells, particularly in the 4 kHz region (due to resonance properties of the ear canal and increased cochlear metabolism at this frequency)
- Audiogram pattern: Characteristic notch at 4000 Hz (4 kHz) with recovery at 8 kHz
- Temporary threshold shift (TTS): reversible loss after brief loud noise exposure — warning sign
- Permanent threshold shift (PTS): irreversible after chronic exposure or acoustic trauma
- Tinnitus in NIHL: high-pitched (4–8 kHz), bilateral, constant, often worse immediately after noise exposure
- Prevention: Hearing protection (earmuffs or earplugs), workplace noise controls (exposure limit 85 dB over 8 hours — OSHA), audiometric surveillance
🔄 Section 9 — Connections to Related Topics
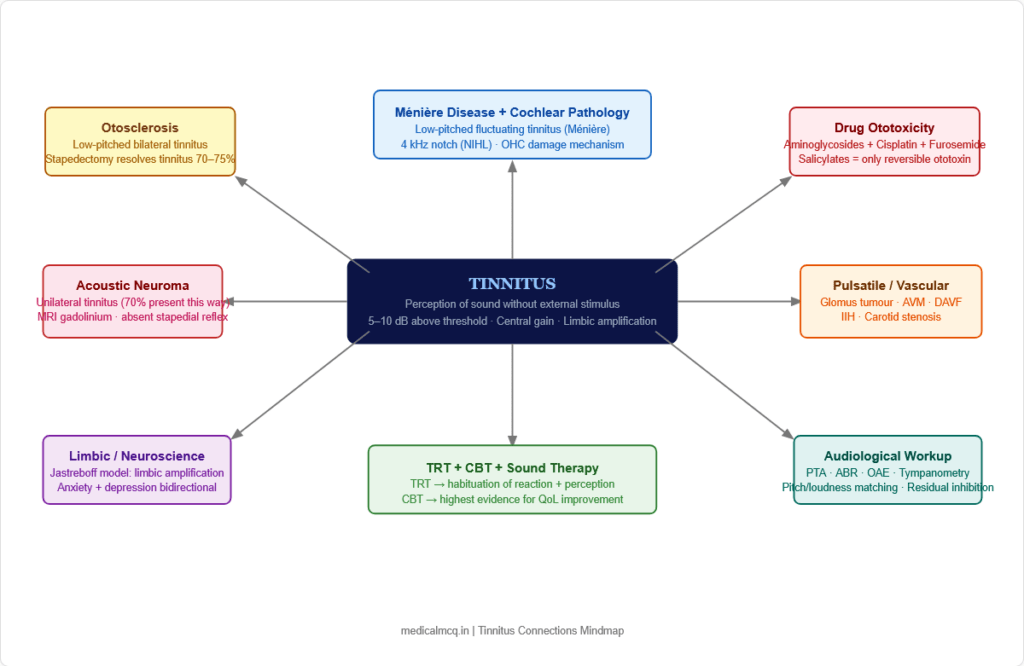
→ Ménière Disease — Endolymphatic hydrops drives fluctuating low-frequency tinnitus. The tinnitus is a cardinal symptom of the diagnostic triad (vertigo, tinnitus, SNHL). Understanding tinnitus pathophysiology explains why the hearing and tinnitus fluctuate together with endolymph pressure changes.
→ Acoustic Neuroma (Vestibular Schwannoma) — Unilateral tinnitus is the first symptom in the majority of cases. The tumour compresses CN VIII → reduces afferent input → central gain increase → tinnitus. This is why MRI is mandatory for unilateral persistent tinnitus.
→ Otosclerosis — Abnormal bone deposition in the otic capsule fixes the stapes → CHL → and cochlear otosclerosis causes additional SNHL. Tinnitus accompanies both forms. Stapedectomy addresses the conductive component and significantly reduces tinnitus.
→ Presbycusis — Age-related SNHL affects OHCs first, primarily at high frequencies. Presbycusis is the most common cause of bilateral high-frequency tinnitus in the elderly. The management is hearing aids (amplification reduces central gain and provides sound enrichment).
→ Drug Ototoxicity — Multiple drug classes damage OHCs (aminoglycosides, cisplatin, loop diuretics, quinine, high-dose aspirin). Understanding ototoxicity monitoring protocols (serial audiometry, OAE monitoring during aminoglycoside therapy) is essential in clinical practice.
→ Vascular Pathology — Pulsatile tinnitus is a presentation of paragangliomas (glomus tumours), AVMs, carotid stenosis, IIH, and carotid-cavernous fistula. The connection between vascular surgery and ENT is tested regularly.
→ Idiopathic Intracranial Hypertension (IIH) — Bilateral pulsatile tinnitus in a young obese woman should always trigger consideration of IIH. The elevated intracranial pressure transmits pulsatile venous pressure waves to the cochlea via the meninges.
→ Neurology — Acoustic neuromas are posterior fossa tumours; large ones cause cerebellar signs, CN VI palsy (from raised ICP), facial nerve involvement. The connection between otological and neurological symptoms is a core exam area.
🎯 High-Yield Exam Facts
🔴 Tinnitus with a 4 kHz audiometric notch = Noise-Induced Hearing Loss (NIHL) The 4 kHz notch is pathognomonic of NIHL. The notch occurs due to the resonance frequency of the external ear canal amplifying sounds at ~3–4 kHz, combined with intense metabolic activity of OHCs at this region making them most vulnerable to acoustic trauma.
🔴 Unilateral persistent tinnitus = MRI with gadolinium until acoustic neuroma excluded Any patient with unexplained unilateral tinnitus needs gadolinium-enhanced MRI. Acoustic neuroma presents with unilateral tinnitus + SNHL + absent stapedial reflex.
🔴 Pulsatile tinnitus synchronising with heartbeat = vascular cause until proven otherwise The list: glomus tumour, AVM, DAVF, carotid stenosis, IIH, high jugular bulb. Order CT/MR angiography. Do NOT biopsy a red mass behind the eardrum without imaging first.
🔴 Acoustic neuroma (vestibular schwannoma): tinnitus + unilateral SNHL + absent stapedial reflex + ABR latency increase The stapedial reflex is absent even with mild SNHL in acoustic neuroma — this is disproportionate to the degree of hearing loss and is a red flag.
🔴 Ototoxic drugs causing tinnitus + SNHL: Aminoglycosides + Cisplatin + Loop diuretics + High-dose salicylates + Quinine The synergistic ototoxicity of aminoglycosides + furosemide is particularly dangerous. High-dose aspirin causes reversible tinnitus (OHC electromotility inhibition via prestin).
🔴 Jastreboff model: Tinnitus = neural signal + limbic emotional reaction + autonomic response. TRT treats the REACTION, not the signal. This is the basis of TRT. The directive counselling component removes fear (limbic deactivation), and sound therapy reduces auditory contrast (reduces central gain). Habituation is the goal.
🟠 Tinnitus in otosclerosis: low-pitched, bilateral; absent stapedial reflex; Carhart notch at 2 kHz; Paracusis Willisii; Schwartze sign Stapedectomy resolves tinnitus in ~70–75% of cases.
🟠 Ménière disease tinnitus: low-pitched, roaring, unilateral, fluctuating — correlates with endolymphatic hydrops SP/AP ratio >0.4 on ECoChG supports Ménière diagnosis. The tinnitus fluctuates with episodes.
🟠 Tinnitus loudness matching typically shows only 5–10 dB above hearing threshold — yet perceived as very loud This central amplification phenomenon explains why even very soft signals (tinnitus) can be severely disabling. It also means tinnitus cannot be masked easily by raising background volume — the brain adjusts.
🟠 Cochlear implants suppress tinnitus in the vast majority of profoundly deaf patients who receive them Electrical stimulation via the implant provides sufficient afferent input to reduce central gain → tinnitus suppression. This is the most dramatic evidence that central gain is the key mechanism.
🟡 CBT has the highest level of evidence for improving quality of life in tinnitus — not loudness reduction, but impact reduction Multiple Cochrane reviews confirm CBT improves emotional and functional response to tinnitus. It does not reduce the loudness — but patients learn to respond to tinnitus differently.
🟡 Residual inhibition — after a masking noise is removed, tinnitus is temporarily suppressed This phenomenon confirms the central auditory system modulates tinnitus and predicts response to sound therapy.
🟡 Somatic tinnitus (modulated by jaw movement or head/neck movement) suggests TMJ dysfunction or cervicogenic origin Asking patients whether moving their jaw, pressing their teeth, or turning their neck changes the tinnitus is a simple but important clinical test. Positive = somatic tinnitus → non-audiological referral.
🟡 IIH-associated tinnitus: bilateral pulsatile + papilloedema + young obese woman + normal MRI → LP shows elevated opening pressure Treatment: Acetazolamide, weight loss, LP shunting for refractory cases. The tinnitus resolves with ICP normalisation.
🧠 Mnemonics & Memory Tricks
“VASCULAR PULSED” — Causes of Pulsatile Tinnitus V = Venous hum (jugular vein) A = AVM (arteriovenous malformation) S = Stenosis of carotid artery C = Carotid-cavernous fistula U = Upper jugular bulb (high/dehiscent) L = Low resistance states (anaemia, pregnancy, thyrotoxicosis) A = Aneurysm (ICA) R = Raised ICP (IIH)
P = Paraganglioma (Glomus tumour) ★ most important U = Unusual bone channels (fibrous dysplasia) L = Lesions of dura (DAVF — dural AV fistula) S = Systemic high-output states E = Ectatic arteries D = Dehiscent carotid canal
💡 Pro tip: Remember “PAR” for the three most testable pulsatile tinnitus causes = Paraganglioma, AVM, Raised ICP (IIH).
“AMINO-CISS-LOOP-QUIN-SALI” — Ototoxic Drugs AMINOglycosides — permanent OHC damage CISSplatin — permanent, basal turn first LOOP diuretics — stria vascularis, usually reversible QUINine + antimalarials — partially reversible SALIcylates (high-dose) — fully reversible (prestin inhibition)
💡 Pro tip: “The ototoxic quintet” = Aminoglycosides, Cisplatin, Loop diuretics, Quinine, Salicylates. The only reversible one at therapeutic doses is salicylates. Aminoglycoside + furosemide = synergistic — never combine without monitoring.
“LOW PITCH = MÉNIÈRE / OTOSCLEROSIS; HIGH PITCH = NIHL / ACOUSTIC NEUROMA”
Low-pitched tinnitus: Ménière (roaring, fluctuating) + Otosclerosis (bilateral, roaring) High-pitched tinnitus: NIHL (4–8 kHz, bilateral) + Acoustic neuroma (unilateral, any pitch but often high)
💡 Pro tip: “Ménière is low, NIHL is high.” This simple rule differentiates the most common tinnitus-associated conditions by the quality of the sound described.
“TRT = Two Rs: Retrain the Reaction, not the Ringing”
TRT works by habituating the limbic REACTION (fear, distress, anxiety) to the tinnitus signal — NOT by eliminating the tinnitus sound itself. = Directive counselling (removes fear → limbic deactivation) + Sound therapy (reduces auditory contrast → reduces central gain → facilitates habituation)
💡 Pro tip: If an exam question asks “what does TRT aim to achieve?” — the answer is “habituation” not “cure” or “masking.”
⚠️ Common Mistakes Students Make on This Topic
❌ Mistake: “Tinnitus always indicates serious underlying pathology” ✅ Reality: The vast majority of tinnitus (especially bilateral, high-pitched, in noise-exposed individuals) is benign and related to cochlear OHC damage from noise or aging. The red flags for serious pathology are: UNILATERAL tinnitus, PULSATILE tinnitus, tinnitus with neurological signs, and tinnitus with rapidly progressive SNHL. 📝 How this gets tested: “A 55-year-old factory worker has bilateral high-pitched tinnitus with a 4 kHz audiogram notch — what investigation is mandatory?” — PTA and occupational history is the answer; not MRI (which would be needed for unilateral tinnitus).
❌ Mistake: “Objective tinnitus can be heard by the patient only, like subjective tinnitus” ✅ Reality: Objective tinnitus is generated by a physical sound source within the body (vascular turbulence, muscle myoclonus) and CAN be heard by the examiner with a stethoscope placed near the ear. It is always pulsatile or rhythmic. Subjective tinnitus (95% of cases) is generated by abnormal neural activity and cannot be heard by anyone else. 📝 How this gets tested: “A pulsatile sound in a patient’s ear is also audible to the examiner with a stethoscope — classification?” = Objective tinnitus. “What common ENT condition causes objective tinnitus?” = Glomus jugulare/tympanicum, AVM, palatal myoclonus.
❌ Mistake: “Stapedectomy cures tinnitus in ALL patients with otosclerosis” ✅ Reality: Stapedectomy resolves or significantly reduces tinnitus in approximately 70–75% of otosclerosis patients. It does NOT guarantee tinnitus elimination. In a small percentage, tinnitus can worsen post-operatively. The surgery primarily addresses the conductive hearing loss; the tinnitus benefit is secondary. 📝 How this gets tested: “A patient with otosclerosis undergoes stapedectomy — what is the expected outcome regarding tinnitus?” = Improvement in ~70–75%, not 100%. Tinnitus may persist in some patients.
❌ Mistake: “Tinnitus loudness matching reveals very loud sounds (e.g., 60–70 dB HL)” ✅ Reality: Tinnitus, when loudness-matched, is typically only 5–10 dB above the patient’s hearing threshold — it is acoustically very soft. The severe impact of tinnitus is NOT due to its acoustic intensity but due to central amplification and the limbic/emotional response it generates. This is the fundamental paradox of tinnitus and the basis of the Jastreboff model. 📝 How this gets tested: “A tinnitus patient reports their ringing is extremely loud. Loudness matching shows the tinnitus corresponds to a sound 8 dB above their hearing threshold. What does this tell us?” = Tinnitus distress is driven by central limbic amplification, not acoustic intensity — the basis of TRT and CBT being the primary treatments.
❌ Mistake: “High-dose aspirin causes permanent ototoxicity” ✅ Reality: High-dose salicylates (>6–8 g/day) cause tinnitus and hearing loss by inhibiting prestin (the OHC motor protein) — but this effect is FULLY REVERSIBLE upon stopping the drug. This contrasts with aminoglycosides and cisplatin, which cause permanent OHC damage. The clinical relevance: patients on high-dose aspirin (e.g., in older rheumatology regimens) who develop tinnitus — stop or reduce the aspirin, and it resolves. 📝 How this gets tested: Comparing ototoxic drugs: “Which of the following ototoxic agents causes REVERSIBLE tinnitus?” = Salicylates (aspirin). All others (aminoglycosides, cisplatin) = largely irreversible.
📝 5 Practice MCQs — Test Yourself Now
Q1: A 48-year-old man presents with a 6-month history of unilateral right-sided tinnitus and progressive right-sided hearing loss. Pure tone audiometry shows right-sided SNHL with poor speech discrimination. Acoustic reflex testing reveals an absent stapedial reflex on the right side despite only mild hearing loss. What is the most likely diagnosis and the investigation of choice?
- A. Ménière disease; Electrocochleography (ECoChG)
- B. Noise-induced hearing loss; Repeat audiogram in 6 months
- C. Acoustic neuroma (vestibular schwannoma); MRI with gadolinium of internal auditory canals
- D. Otosclerosis; CT temporal bones
✅ Answer: C. Acoustic neuroma; MRI with gadolinium of IACs
Why correct: The combination of UNILATERAL tinnitus + progressive unilateral SNHL + absent stapedial reflex DISPROPORTIONATE to the degree of hearing loss is the classic triad of acoustic neuroma (vestibular schwannoma). The absent stapedial reflex with only mild SNHL is particularly suspicious — in cochlear hearing loss, reflex thresholds are elevated but the reflex is present (loudness recruitment); in retrocochlear pathology (acoustic neuroma), the reflex is absent even with mild loss because the efferent loop of the reflex arc (CN VII) remains intact but the afferent (CN VIII) is dysfunctional. MRI with gadolinium of the brain and internal auditory canals is the gold standard investigation — it will show an enhancing mass in the IAC or CPA angle.
Why A is wrong: Ménière disease typically causes LOW-pitched fluctuating tinnitus, episodic vertigo, and LOW-frequency SNHL — not progressive unilateral SNHL with absent stapedial reflex. ECoChG is used to confirm Ménière (SP/AP ratio). Why B is wrong: NIHL causes bilateral high-frequency SNHL with a 4 kHz notch — not progressive unilateral SNHL. Stapedial reflexes are present in NIHL. Why D is wrong: Otosclerosis causes conductive or mixed HL with an air-bone gap and absent stapedial reflex — but audiometry would show a Carhart notch (2 kHz) and an air-bone gap, not purely sensorineural loss with poor speech discrimination.
Exam tip: The triad of unilateral tinnitus + unilateral SNHL + absent stapedial reflex = acoustic neuroma until proven otherwise. MRI with gadolinium is mandatory.
Q2: A 35-year-old woman presents with pulsatile tinnitus in her right ear that she describes as a “whooshing sound” synchronising with her heartbeat. On examination, you notice a pinkish-red pulsatile mass visible behind the right tympanic membrane in the inferior quadrant. What is the correct immediate management?
- A. Proceed with fine needle aspiration biopsy of the mass to obtain a tissue diagnosis
- B. Immediate CT temporal bones and CT angiography before any intervention; do not biopsy
- C. Perform myringotomy and drain any fluid from the middle ear
- D. Start intravenous antibiotics for suspected acute otitis media with effusion
✅ Answer: B. Immediate CT temporal bones and CT angiography before any intervention; do not biopsy
Why correct: A pulsatile red mass behind the tympanic membrane in the inferior quadrant (hypotympanum) is a glomus tympanicum tumour (paraganglioma) until proven otherwise. These are highly vascular tumours arising from the paraganglionic tissue in the middle ear or jugular bulb. Blindly biopsying this mass without imaging could cause catastrophic haemorrhage because these tumours are fed by prominent arterial branches (ascending pharyngeal artery, inferior tympanic artery). Imaging first — CT temporal bones (bony detail) and CT angiography (vascular supply) — provides diagnosis AND surgical planning. Treatment is surgical resection ± pre-operative embolisation.
Why A is wrong: Blind biopsy of a vascular middle ear mass is potentially life-threatening. This is one of the most important “do NOT do” rules in ENT — never biopsy a red mass behind the tympanic membrane without prior imaging. Why C is wrong: Myringotomy in the presence of a vascular mass would cause catastrophic haemorrhage. This is absolutely contraindicated. Why D is wrong: A pulsatile mass is not consistent with acute otitis media with effusion (glue ear). AOM/effusion causes a dull, retracted, non-pulsatile TM; there is no red pulsatile mass. Antibiotics have no role here.
Exam tip: “Red pulsatile mass behind tympanic membrane” = Glomus tympanicum = DO NOT biopsy = IMAGING FIRST. This is one of the highest-yield ENT examination rules and appears regularly in clinical scenario MCQs.
Q3: A 28-year-old woman who is 30 weeks pregnant presents with bilateral pulsatile tinnitus and headaches. On fundoscopy, bilateral papilloedema is found. MRI brain is normal with no mass lesion. What is the most likely diagnosis, and what investigation will confirm it?
- A. Bilateral acoustic neuromas (NF2); MRI with gadolinium of IACs
- B. Idiopathic Intracranial Hypertension (IIH); Lumbar puncture to measure opening pressure
- C. Bilateral Ménière disease; Electrocochleography (ECoChG)
- D. Carotid-cavernous fistula; Orbital ultrasound
✅ Answer: B. Idiopathic Intracranial Hypertension (IIH); Lumbar puncture
Why correct: This is the classic presentation of IIH (pseudotumour cerebri): young woman (pregnancy is a risk factor, as is obesity) with bilateral pulsatile tinnitus, headaches, and papilloedema, with a normal MRI. The pulsatile tinnitus in IIH is caused by elevated intracranial venous pressure being transmitted to the transverse/sigmoid sinuses adjacent to the temporal bone → pulsatile sound perceived via the cochlea. The normal MRI excludes a structural cause of raised ICP. Lumbar puncture confirms the diagnosis by demonstrating elevated cerebrospinal fluid opening pressure (>250 mm H₂O in the lateral decubitus position) with normal CSF composition.
Why A is wrong: Bilateral acoustic neuromas suggest NF2 (neurofibromatosis type 2), but these cause bilateral SNHL with masses visible on MRI gadolinium enhancement in the IACs — the MRI is normal here. NF2 does not cause papilloedema or pulsatile tinnitus. Why C is wrong: Bilateral Ménière disease is rare and presents with episodic vertigo + LOW-pitched fluctuating tinnitus — not bilateral pulsatile tinnitus with papilloedema. ECoChG would show SP/AP >0.4 in Ménière. Why D is wrong: Carotid-cavernous fistula presents with pulsatile proptosis, chemosis, conjunctival injection, and an orbital bruit — not bilateral pulsatile tinnitus with papilloedema.
Exam tip: IIH diagnostic criteria: bilateral pulsatile tinnitus + headache + papilloedema + normal MRI + elevated ICP on LP (>250 mm H₂O) + normal CSF composition. Young obese women. Treat with acetazolamide + weight loss.
Q4: A patient on gentamicin therapy for a severe gram-negative infection is being monitored with serial audiometry. After 7 days of treatment, the audiogram shows a new bilateral SNHL at 8000 Hz that was not present at baseline. The clinical team asks whether to continue gentamicin. What is the correct advice, and which additional intervention should be considered to reduce further cochlear damage?
- A. Continue gentamicin at the same dose; SNHL at 8000 Hz is an age-related change
- B. Discontinue gentamicin immediately; switch to a non-ototoxic alternative if clinically possible; high frequencies are affected first — this is early cochleotoxicity
- C. Reduce the dose of gentamicin by half; co-administer furosemide to increase renal clearance
- D. Continue gentamicin and add oral aspirin 300 mg to protect cochlear blood flow
✅ Answer: B. Discontinue gentamicin immediately; switch to a non-ototoxic alternative if clinically possible
Why correct: Aminoglycoside ototoxicity causes progressive OHC damage starting at the base of the cochlea (high frequencies) and moving towards the apex (low frequencies) as damage progresses. The 8000 Hz (8 kHz) notch appearing after 7 days is early cochleotoxicity — a warning sign that irreversible cochlear damage is occurring and will progress to lower frequencies (speech frequencies) if the drug is continued. Serial audiometry is done precisely to catch this early warning and allow drug discontinuation before speech-frequency hearing is affected. If a non-ototoxic alternative exists (e.g., a beta-lactam, fluoroquinolone), switch immediately. If gentamicin is absolutely necessary for survival (e.g., no alternative effective antibiotic), the risk-benefit balance must be carefully weighed and trough/peak drug levels optimised.
Why A is wrong: A new high-frequency SNHL appearing after 7 days of aminoglycoside therapy is NOT age-related — it is aminoglycoside cochleotoxicity until proven otherwise. Age-related SNHL is gradual and bilateral, not appearing acutely over 7 days. Why C is wrong: Reducing the dose may slow progression but does not reverse established OHC damage. More critically, co-administering furosemide is CONTRAINDICATED — aminoglycoside + loop diuretic = synergistic ototoxicity, dramatically increasing cochlear damage risk. Why D is wrong: There is no evidence that aspirin protects against aminoglycoside cochleotoxicity — in fact, high-dose aspirin is itself ototoxic. Adding aspirin makes no clinical sense here.
Exam tip: Aminoglycoside ototoxicity: damages base first → high frequencies first → monitoring with 8 kHz audiometry is most sensitive → if 8 kHz loss appears, discontinue if possible → furosemide co-administration is specifically contraindicated (synergistic toxicity).
Q5: A 62-year-old man with bilateral tinnitus and mild bilateral SNHL has been using a white noise masker for 6 months with limited benefit. He scores 64/100 on the Tinnitus Handicap Inventory (severe grade). He reports significant anxiety and sleep disturbance. He is referred for Tinnitus Retraining Therapy (TRT). A colleague suggests that TRT aims to eliminate the tinnitus completely. Is this correct, and what is the actual goal of TRT?
- A. Correct — TRT eliminates tinnitus by retraining the auditory cortex to no longer generate the signal
- B. Incorrect — TRT aims to achieve habituation of both the perception of and the reaction to tinnitus, making it no longer bothersome even if still audible
- C. Incorrect — TRT only works by masking tinnitus with sound; complete masking is the endpoint
- D. Correct — TRT uses cochlear implant stimulation to permanently suppress the tinnitus signal
✅ Answer: B. Incorrect — TRT aims to achieve habituation of both perception and reaction, not elimination
Why correct: This question directly tests understanding of the Jastreboff neurophysiological model and TRT goals. TRT does NOT aim to eliminate tinnitus — it aims to achieve habituation. Jastreboff distinguishes between habituation of REACTION (the emotional/autonomic distress response — achieved first and most important) and habituation of PERCEPTION (becoming unaware of the tinnitus signal — achieved later). TRT uses two tools: (1) Directive counselling — to reclassify tinnitus as a meaningless, non-threatening signal, deactivating the limbic response; and (2) Sound therapy — broadband sound at or just below the masking level (“mixing point”) to reduce auditory contrast. The endpoint is that patients no longer notice their tinnitus most of the time and are no longer distressed by it when they do — even though the underlying auditory signal may still be present. This patient’s high THI score (64 = severe) and anxiety make him an excellent candidate for TRT combined with CBT.
Why A is wrong: TRT does not eliminate the neural signal generating tinnitus — it habituates the system’s RESPONSE to it. The auditory cortex may continue to generate the signal, but the limbic and autonomic systems no longer react to it. Why C is wrong: Sound therapy in TRT is specifically set BELOW the masking level (at the “mixing point” — where the sound partially blends with tinnitus but does not completely mask it). Complete masking is NOT the goal of TRT; it is the approach of simple masking therapy, which is less effective. Why D is wrong: TRT does not use cochlear implants. Cochlear implants ARE used in specific patients with profound SNHL, where the electrical stimulation incidentally suppresses tinnitus — but this is a different treatment for a different indication.
Exam tip: TRT goals = HABITUATION (not cure, not masking). Two components: directive counselling + sound therapy at mixing point. Duration: 12–24 months. The Jastreboff model explains WHY it works: by deactivating the limbic emotional response, the tinnitus signal loses its threatening quality and becomes habituated — like a background sound we no longer consciously hear.
📚 References
📖 Scott-Brown’s Otorhinolaryngology, Head and Neck Surgery — Gleeson et al. | Chapter on Tinnitus
📖 Cummings Otolaryngology Head and Neck Surgery — Flint et al. | Chapter 162: Tinnitus and Hyperacusis
📖 Clinical Audiology: An Introduction — Stach | Chapter on Tinnitus Assessment
📖 Jastreboff PJ, Hazell JWP — Tinnitus Retraining Therapy | Cambridge University Press
📖 Bhatt JM, Lin HW, Bhattacharyya N — Prevalence, Severity, Exposures and Treatment Patterns of Tinnitus in the United States | JAMA Otolaryngol Head Neck Surg 2016
📖 National Institute for Health and Care Excellence (NICE) — Tinnitus: Assessment and Management | NG155, 2020
🚀 Keep Practising — You Are Not Done Yet
Tinnitus questions in NEET PG and USMLE are almost always embedded within broader ENT clinical scenarios — a patient with Ménière disease happens to mention tinnitus, a man with noise-induced HL has the characteristic 4 kHz notch, a woman with a pulsatile sound behind her eardrum has a glomus tumour. The ability to extract the relevant tinnitus features from a clinical vignette and map them to the correct diagnosis is what separates high scorers.
Practise with clinical-scenario MCQs — not just definition-type questions. The patterns become automatic with repetition.
medicalmcq.in has free ENT and Otolaryngology MCQs covering tinnitus, Ménière disease, acoustic neuroma, otosclerosis, and NIHL — all in clinical-scenario format with detailed mechanistic explanations.
