What You Will Learn in This Article
- Trace the complete synthesis of T3 and T4 from iodide uptake through organification, coupling, and secretion
- Explain the HPT axis — TRH, TSH, and all feedback mechanisms
- Describe the transport of thyroid hormones in blood and the critical distinction between total and free hormone
- List all the metabolic, cardiovascular, skeletal, CNS, and haematological actions of thyroid hormones
- Recognise and differentiate the causes, clinical features, investigations, and treatment of hyperthyroidism and hypothyroidism
- Identify the specific clinical syndromes: Graves disease, Hashimoto thyroiditis, thyroid storm, myxoedema coma, sick euthyroid syndrome
- Explain how drugs (PTU, methimazole, amiodarone, lithium, iodide) affect thyroid function
- Understand thyroid cancer — types, features, and treatment
📖 Introduction: Why This Topic Matters in Exams
A 28-year-old woman presents with a 3-month history of weight loss despite increased appetite, palpitations, tremor, heat intolerance, and diarrhoea. Her eyes appear prominent. On examination her thyroid is diffusely enlarged, she is tachycardic with an irregular pulse, and there is a fine tremor of outstretched hands. TSH is suppressed to <0.01 mIU/L and free T4 is markedly elevated. TSH receptor antibodies are positive. She has Graves disease — the most common cause of hyperthyroidism — and every single feature of her presentation follows mechanistically from excess thyroid hormone action. Understanding this pathway is what separates a student who can merely name the conditions from one who can explain why each sign and symptom occurs.
Thyroid hormones are tested across virtually every discipline in NEET PG and USMLE Step 1. In biochemistry: the synthesis pathway, iodine metabolism, and enzyme defects. In physiology: the HPT axis, negative feedback, and the wide-ranging metabolic effects. In endocrinology: the full clinical picture of Graves, Hashimoto, thyroid storm, myxoedema coma, and congenital hypothyroidism. In pharmacology: PTU, methimazole, radioactive iodine, amiodarone, and lithium. In pathology: the autoimmune basis of Graves and Hashimoto, and thyroid cancer classification.
This article covers all of it, systematically and mechanistically.
🔬 Section 1 — Thyroid Hormone Synthesis: From Iodide to T3 and T4
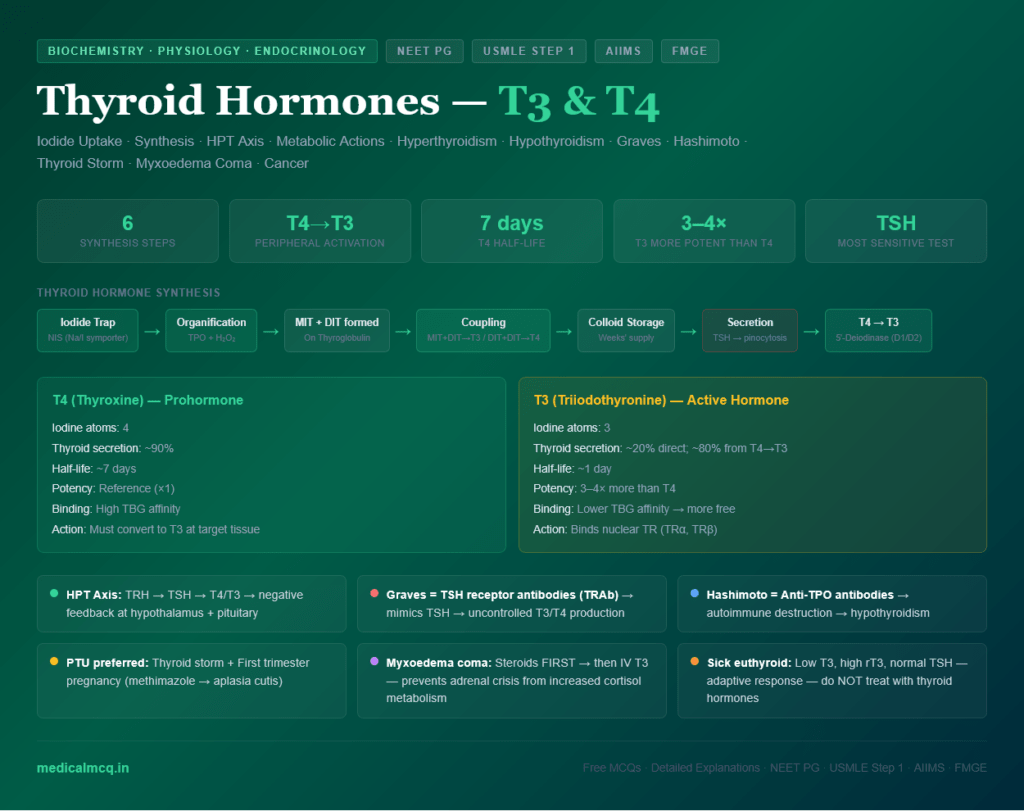
The Chemistry of Thyroid Hormones
Thyroid hormones are iodinated derivatives of tyrosine. They are unique among hormones in requiring an essential trace element — iodine — for their synthesis. The two principal thyroid hormones are:
- Thyroxine (T4): Contains 4 iodine atoms; the major secretory product of the thyroid (90% of secretion)
- Triiodothyronine (T3): Contains 3 iodine atoms; the biologically active form at cellular level; 3–4× more potent than T4; mainly produced by peripheral deiodination of T4
Reverse T3 (rT3): An isomer of T3 produced by alternative deiodination of T4 (removing iodine from the inner rather than outer ring). rT3 is metabolically inactive and is elevated in sick euthyroid syndrome.
Step-by-Step Synthesis
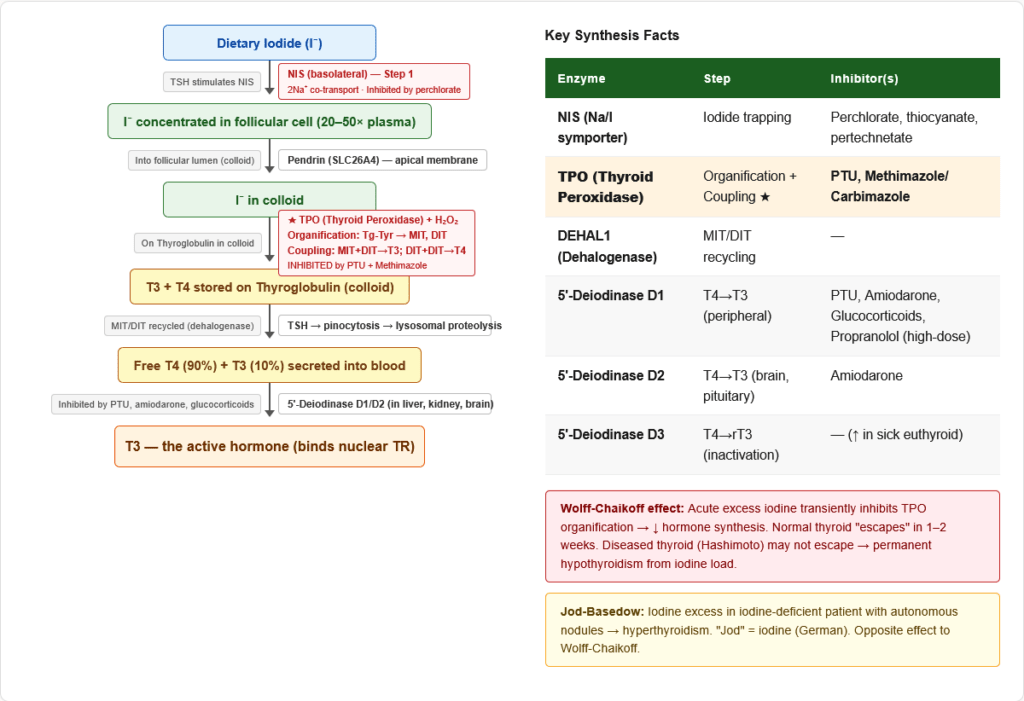
Step 1 — Iodide Uptake (Trapping)
- Iodide (I⁻) from food is actively transported from the blood into thyroid follicular cells against an electrochemical gradient
- Transporter: Sodium-Iodide Symporter (NIS) — co-transports 2 Na⁺ with each I⁻
- NIS is on the basolateral membrane of follicular cells
- The iodide concentration inside the thyroid is 20–50× higher than in plasma
- Stimulated by: TSH
- Inhibited by: Perchlorate (ClO₄⁻), thiocyanate (SCN⁻), pertechnetate (TcO₄⁻) — competitive inhibitors that use the same transporter
- This trapping mechanism is exploited clinically: Radioactive iodine (¹³¹I) is taken up selectively by thyroid tissue → used for thyroid scanning, hyperthyroidism treatment, and thyroid cancer ablation
Step 2 — Oxidation and Organification
- I⁻ is transported across the apical membrane by Pendrin (SLC26A4) into the follicular lumen
- In the follicular lumen, I⁻ is oxidised to reactive iodine (I⁰ or I⁺) by Thyroid Peroxidase (TPO) — an enzyme embedded in the apical membrane — using H₂O₂ as the oxidant
- Reactive iodine is immediately incorporated into tyrosine residues on thyroglobulin (Tg) — a large glycoprotein (660 kDa) secreted by follicular cells into the colloid
- This process is called organification: Tyrosine residues on Tg → iodinated
- Results in:
- Monoiodotyrosine (MIT): One iodine added to tyrosine
- Diiodotyrosine (DIT): Two iodines added to tyrosine
- Inhibited by: PTU (propylthiouracil), methimazole/carbimazole — these are the primary antithyroid drugs; they inhibit TPO → block organification
Step 3 — Coupling Reaction
- Within the thyroglobulin molecule, MIT and DIT are coupled by TPO to form the iodothyronines:
- MIT + DIT → T3 (one 3-carbon iodotyrosine: the 3-iodo form)
- DIT + DIT → T4 (two iodinations coupled: the 4-iodo form)
- The coupling reaction is also catalysed by TPO and requires H₂O₂
- The proportion of T4 > T3 in the coupled product (approximately 10:1 T4:T3)
- Inhibited by: PTU and methimazole (both inhibit coupling via TPO inhibition)
Step 4 — Storage
- Coupled iodothyronines (T3, T4) remain covalently linked to thyroglobulin in the follicular colloid — this is the unique storage form of thyroid hormones
- The colloid can store several weeks’ worth of thyroid hormones — this is why antithyroid drugs take weeks to reduce circulating levels, and why an iodine load can acutely exacerbate hyperthyroidism (Jod-Basedow effect)
Step 5 — Resorption and Secretion
- TSH stimulates pinocytosis of colloid droplets back into the follicular cell
- Lysosomes fuse with colloid droplets → proteolysis of thyroglobulin by lysosomal proteases (cathepsins) → releases T4, T3, MIT, DIT
- MIT and DIT are deiodinated within the cell by iodotyrosine dehalogenase (DEHAL1/IYD) → tyrosine + iodide recycled (iodide conservation)
- Free T4 and T3 diffuse across the basolateral membrane into the bloodstream
- Transporter: MCT8 (Monocarboxylate transporter 8) — mutations cause Allan-Herndon-Dudley syndrome (severe X-linked intellectual disability with abnormal thyroid function tests)
Step 6 — Peripheral Conversion: T4 → T3
- T4 is largely a prohormone — it must be converted to T3 for biological activity
- 5′-Deiodinase (Type I and Type II) removes iodine from the outer ring of T4 → T3 (the active hormone)
- Type I (D1): Liver, kidney, thyroid, muscle — high-capacity enzyme; provides ~80% of circulating T3
- Type II (D2): Brain, pituitary, brown adipose tissue — local T3 for these tissues
- Type III (D3): Placenta, brain — inactivates T4 → rT3 and T3 → T2; protects foetus from excess maternal thyroid hormones
- Inhibited by: PTU (ONLY Type I deiodinase) — this is why PTU is preferred over methimazole in thyroid storm (it both blocks synthesis AND peripheral conversion of T4 to T3). Amiodarone, high-dose propranolol, glucocorticoids, and severe illness also inhibit peripheral conversion.
Summary: Key Enzymes in Thyroid Hormone Synthesis
| Enzyme | Step | Location | Inhibited By |
|---|---|---|---|
| NIS (Na/I symporter) | Iodide trapping | Basolateral membrane | Perchlorate, thiocyanate |
| Pendrin (SLC26A4) | Iodide transport to colloid | Apical membrane | — |
| Thyroid Peroxidase (TPO) | Organification + Coupling | Apical membrane | PTU, Methimazole |
| Iodotyrosine Dehalogenase | MIT/DIT recycling | Intracellular | — |
| 5′-Deiodinase Type I | T4 → T3 (peripheral) | Liver, kidney | PTU, Amiodarone |
| 5′-Deiodinase Type II | T4 → T3 (local) | Brain, pituitary | Amiodarone |
| 5′-Deiodinase Type III | T4 → rT3 (inactivation) | Placenta, brain | — |
⚙️ Section 2 — The HPT Axis: Regulation of Thyroid Hormones
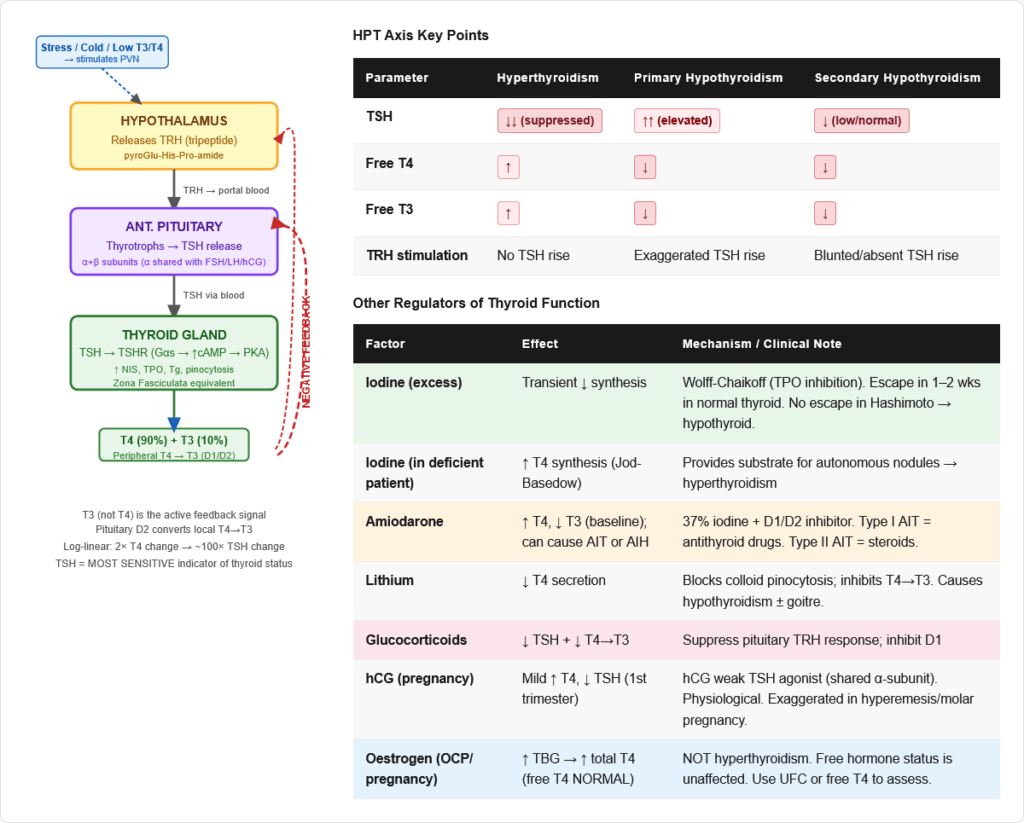
The Three-Tier Hierarchy
Level 1 — Hypothalamus:
- Releases Thyrotropin-Releasing Hormone (TRH) from the paraventricular nucleus
- TRH is a tripeptide (pyroGlu-His-Pro-amide) — smallest classical hypothalamic releasing hormone
- TRH travels via the portal system to the anterior pituitary
- Stimulated by: cold exposure, low circulating T3/T4
- Inhibited by: high T3/T4 (negative feedback), dopamine, somatostatin, glucocorticoids
Level 2 — Anterior Pituitary (Thyrotrophs):
- TRH binds → stimulates TSH (Thyroid-Stimulating Hormone) release
- TSH is a glycoprotein (α + β subunits; the α subunit is shared with FSH, LH, and hCG)
- TSH is the most important regulator of thyroid function
- Inhibited by: high T3/T4 (dominant negative feedback), somatostatin, dopamine, glucocorticoids
- T3 (not T4) acts at the pituitary to suppress TSH — pituitary D2 converts local T4 → T3 for feedback
Level 3 — Thyroid Gland:
- TSH binds to TSH receptor (TSHR) on follicular cell basolateral membrane
- TSHR is a G-protein coupled receptor → stimulates Gαs → activates adenylyl cyclase → ↑ cAMP → PKA activation
- At higher TSH levels: also activates Gαq → phospholipase C → DAG + IP3 → PKC
- TSH actions: stimulates all steps of thyroid hormone synthesis (NIS, TPO, Tg synthesis, pinocytosis, T4 secretion), promotes thyroid cell growth (thyroid hypertrophy/hyperplasia)
Negative Feedback — The Core Regulatory Principle
Thyroid hormone feedback is predominantly at the level of the pituitary:
- T3 (from peripheral conversion of T4) binds to thyroid hormone receptor (TR) in pituitary thyrotrophs → suppresses POMC gene transcription → ↓ TSH
- This is why TSH is the most sensitive indicator of thyroid status:
- Even small changes in free T4 produce large reciprocal changes in TSH
- TSH is LOW in hyperthyroidism (excess T3/T4 suppresses pituitary)
- TSH is HIGH in primary hypothyroidism (insufficient T3/T4 → inadequate pituitary suppression)
- Log-linear relationship: A 2-fold change in free T4 produces a ~100-fold change in TSH
Other Regulators
| Factor | Effect on Thyroid |
|---|---|
| Iodine (Wolff-Chaikoff effect) | Acute excess iodine → TRANSIENTLY inhibits TPO → reduces hormone synthesis. Normal thyroid “escapes” after 1–2 weeks. Diseased thyroid (Hashimoto) may fail to escape → hypothyroidism. |
| Jod-Basedow effect | Iodine excess in iodine-deficient patient → triggers hyperthyroidism (provides substrate for pre-formed autonomous nodules) |
| Pregnancy | hCG has weak TSH-like activity (shares α-subunit of TSH) → mild stimulation of thyroid in first trimester → mild ↑ T4, ↓ TSH in normal pregnancy |
| Glucocorticoids | Suppress TSH release; inhibit peripheral T4→T3 conversion |
| Dopamine, somatostatin | Inhibit TSH release (used to test pituitary reserve) |
🔬 Section 3 — Transport and Distribution
Binding Proteins in Blood
- Thyroid-Binding Globulin (TBG): Binds ~70% of circulating T4 and T3 with highest affinity; synthesised in liver
- Transthyretin (TTR / Prealbumin): Binds ~15–20% of T4 (lower affinity than TBG)
- Albumin: Binds ~10% — lowest affinity, highest capacity
Only the FREE fraction is biologically active:
- Free T4: ~0.02–0.04% of total T4
- Free T3: ~0.3–0.5% of total T3
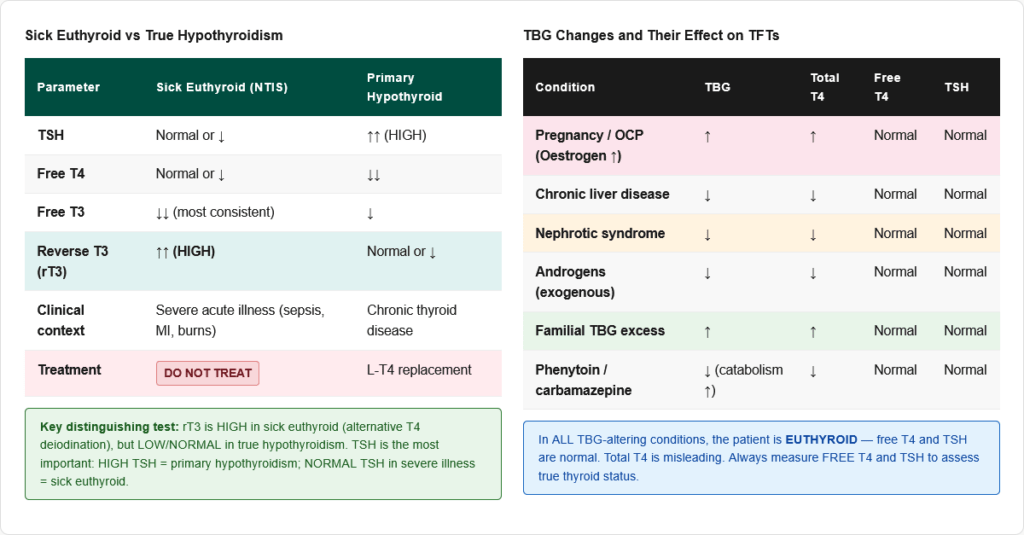
Clinical implications of TBG changes:
| Condition | TBG | Total T4/T3 | Free T4/T3 | TSH | Clinical Status |
|---|---|---|---|---|---|
| Pregnancy / OCP (oestrogen) | ↑ | ↑ | Normal | Normal | Euthyroid |
| Liver disease (chronic) | ↓ | ↓ | Normal | Normal | Euthyroid |
| Nephrotic syndrome | ↓ | ↓ | Normal | Normal | Euthyroid |
| Androgens | ↓ | ↓ | Normal | Normal | Euthyroid |
| Familial TBG excess | ↑ | ↑ | Normal | Normal | Euthyroid |
Key rule: Always measure FREE T4 and FREE T3 to assess true thyroid function. Total T4/T3 can be misleading due to TBG changes.
Peripheral Metabolism
Plasma half-life:
- T4: ~7 days (longest of any thyroid hormone — due to high TBG binding)
- T3: ~1 day
Distribution:
- ~80% of circulating T3 comes from peripheral deiodination of T4 (not direct thyroid secretion)
- ~20% of T3 is secreted directly by the thyroid
Excretion:
- Conjugated with glucuronide and sulphate in liver → excreted in bile → enterohepatic recirculation
- Small amount excreted in urine as glucuronide/sulphate conjugates
🔬 Section 4 — Mechanism of Action: How Thyroid Hormones Work
Nuclear Receptor Pathway
T3 (not T4) binds to Thyroid Hormone Receptors (TRα and TRβ) — nuclear receptors that are members of the steroid/nuclear receptor superfamily.
Unlike steroid hormone receptors (which are in the cytoplasm), TRs are already bound to DNA in the nucleus in the apo-receptor form (without hormone), where they actively repress gene transcription by recruiting co-repressors (including HDACs).
When T3 binds:
- Co-repressors are released
- Co-activators are recruited
- Chromatin remodelling occurs
- Target gene transcription is activated or repressed
Thyroid Hormone Response Elements (TREs) in the promoters of target genes mediate the transcriptional effects.
Key Target Genes
| Effect | Mechanism (T3 target gene) |
|---|---|
| ↑ Metabolic rate | ↑ Na⁺/K⁺-ATPase (energy-consuming) |
| ↑ Heart rate / contractility | ↑ β-adrenergic receptors; ↑ Ca²⁺ channels; ↑ myosin heavy chain α |
| ↑ Thermogenesis | ↑ UCP-1 (uncoupling protein) in brown fat; ↑ mitochondrial biogenesis |
| ↑ Protein synthesis | General anabolic effect at physiological levels |
| ↑ Lipolysis | ↑ Hormone-sensitive lipase |
| ↑ Gluconeogenesis | ↑ PEPCK |
| ↑ Bone turnover | ↑ Osteoblast AND osteoclast activity |
| Brain development | Critical for neuronal migration, myelination, synaptogenesis in foetal/neonatal period |
🔬 Section 5 — Physiological Actions of Thyroid Hormones
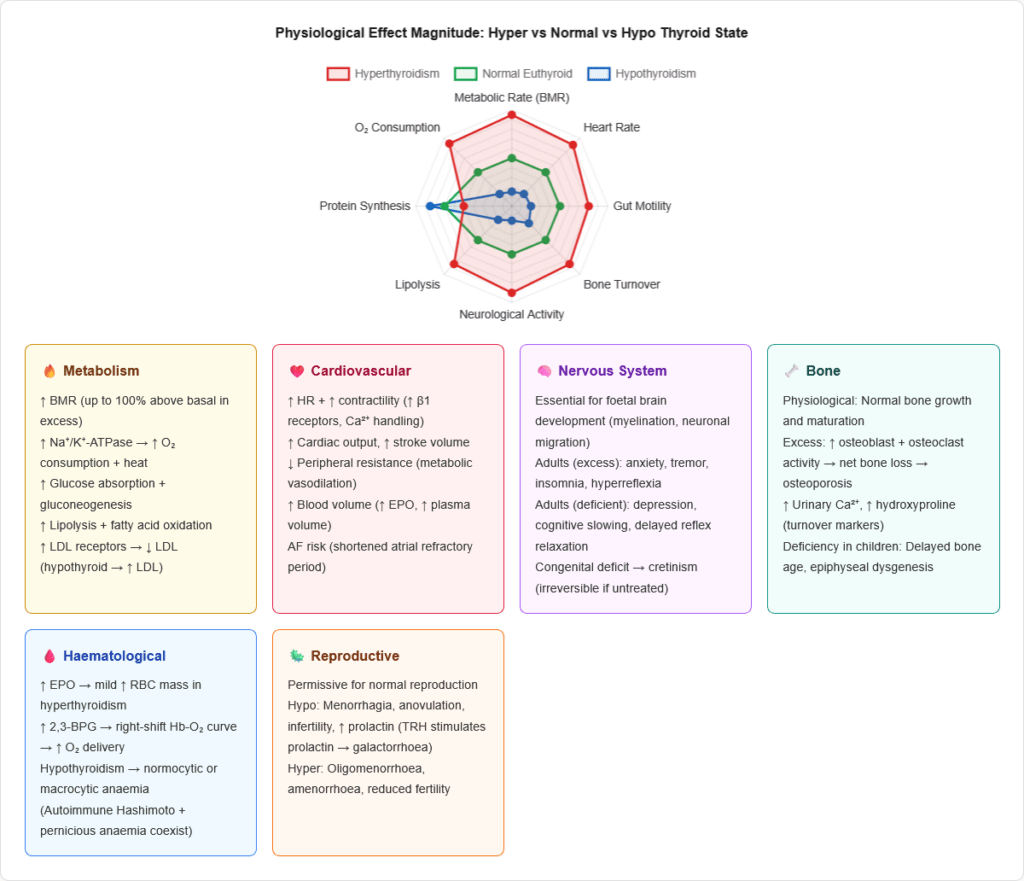
1. Metabolic Effects — The Master Metabolic Regulator
Basal Metabolic Rate (BMR):
- Thyroid hormones are the single most important determinant of BMR
- T3 increases BMR by 60–100% above basal when present at excess levels
- Mechanism: ↑ Na⁺/K⁺-ATPase activity throughout the body → increased ATP consumption → increased O₂ consumption and heat generation
- Hyperthyroidism: High BMR → weight loss, heat intolerance, sweating despite increased food intake
- Hypothyroidism: Low BMR → weight gain, cold intolerance
Carbohydrate metabolism:
- ↑ Intestinal glucose absorption
- ↑ Gluconeogenesis and glycogenolysis (at high levels — counter-regulatory)
- ↑ Peripheral glucose utilisation
- Net: mild hyperglycaemia tendency in hyperthyroidism; frank diabetes can be unmasked
Fat metabolism:
- ↑ Lipolysis (mobilise fatty acids)
- ↑ Fatty acid oxidation
- ↑ LDL receptor expression → ↑ LDL clearance
- Result: Hypothyroidism → ↑ LDL cholesterol → dyslipidaemia, atherosclerosis risk
Protein metabolism:
- Physiological levels: Anabolic (promotes protein synthesis)
- Supraphysiological levels (hyperthyroidism): Catabolic (muscle wasting, negative nitrogen balance)
2. Cardiovascular Effects
| Effect | Mechanism |
|---|---|
| ↑ Heart rate | Direct sinoatrial node stimulation + ↑ β1-adrenergic receptor expression |
| ↑ Cardiac contractility | ↑ Myosin ATPase, Ca²⁺ handling proteins (SERCA2) |
| ↑ Cardiac output | ↑ HR × ↑ SV |
| ↓ Peripheral vascular resistance | Vasodilatation from metabolic heat → ↑ vasodilatory mediators |
| ↑ Blood volume | ↑ Erythropoietin, ↑ plasma volume |
| Atrial fibrillation | Excess TH → SA node instability, shortened refractory period → AF (especially in elderly) |
Hyperthyroidism → High-output state: High HR, wide pulse pressure, bounding pulse, AF Hypothyroidism → Low-output state: Bradycardia, ↑ diastolic BP, narrow pulse pressure, pericardial effusion
3. Nervous System Effects
Central nervous system:
- Thyroid hormones are ESSENTIAL for normal brain development in the foetal and neonatal period
- Foetal thyroid begins functioning at ~12 weeks gestation; maternal T4 crosses placenta before this
- Congenital hypothyroidism → Cretinism if untreated: irreversible intellectual disability, deafness, spasticity, short stature
- The critical window is the first 3 years of life — this is why newborn screening and early treatment are essential
- In adults: Hyperthyroidism → anxiety, irritability, insomnia, fine tremor, hyperreflexia
- Hypothyroidism → depression, cognitive slowing, delayed relaxation phase of deep tendon reflexes (the “hung-up” reflex — pathognomonic)
Peripheral nervous system:
- TH maintains normal nerve conduction
- Hypothyroidism → carpal tunnel syndrome (myxoedematous infiltration of carpal tunnel), peripheral neuropathy
4. Bone and Mineral Metabolism
- Physiological T3 promotes normal bone growth and maturation
- Hyperthyroidism: ↑ both osteoblast and osteoclast activity → net bone loss → osteoporosis; hypercalciuria; ↑ urinary hydroxyproline (bone turnover marker)
- Hypothyroidism in children: Delayed bone age, epiphyseal dysgenesis, stunted growth
5. Haematological Effects
- ↑ EPO → ↑ RBC mass (mild polycythaemia in hyperthyroidism)
- ↑ 2,3-BPG → right-shift of Hb-O₂ curve → better O₂ delivery to tissues
- Hypothyroidism → normocytic anaemia (reduced EPO) or macrocytic anaemia (associated pernicious anaemia in autoimmune hypothyroidism)
6. Gastrointestinal Effects
- Hyperthyroidism: ↑ GI motility → diarrhoea, increased bowel frequency, malabsorption
- Hypothyroidism: ↓ GI motility → constipation, ileus
7. Reproductive Effects
- Thyroid hormones are permissive for normal reproductive function
- Hypothyroidism: Menorrhagia (anovulatory cycles), infertility, hyperprolactinaemia (↑ TRH also stimulates prolactin release)
- Hyperthyroidism: Oligomenorrhoea/amenorrhoea, infertility
🏥 Section 6 — Hyperthyroidism: Excess Thyroid Hormone
Causes
| Cause | Mechanism | Key Features |
|---|---|---|
| Graves disease (most common, ~70–80%) | Autoimmune — TSH receptor antibodies (TRAb/TSIG) stimulate TSHR autonomously | Diffuse goitre, exophthalmos, pretibial myxoedema, acropachy |
| Toxic multinodular goitre (Plummer disease) | Autonomous nodules — somatic TSHR or Gsα mutations → TSH-independent function | Older patients; multiple nodules; no ophthalmopathy |
| Toxic adenoma | Single autonomous nodule with TSHR mutation | Single “hot” nodule on scan; suppressed surrounding tissue |
| Subacute thyroiditis (de Quervain) | Viral-induced inflammation → destructive release of preformed hormones | Painful thyroid; elevated ESR; self-limiting; hypothyroid phase follows |
| Hashimoto thyroiditis (Hashitoxicosis) | Autoimmune destruction → transient release of hormones | Brief hyperthyroid phase before permanent hypothyroidism |
| Factitious hyperthyroidism | Exogenous T4/T3 ingestion | Low Tg; no goitre |
| Struma ovarii | Ectopic thyroid tissue in ovarian teratoma | Rare |
| TSH-secreting pituitary adenoma | High TSH + high T4 | Central hyperthyroidism; TSH inappropriately normal/high |
| hCG-mediated (gestational thyrotoxicosis) | hCG cross-reacts with TSHR in first trimester | Common in molar pregnancy, hyperemesis gravidarum |
| Amiodarone-induced (Type I/II) | Type I: excess iodine → autonomous synthesis; Type II: destructive thyroiditis | Amiodarone contains 37% iodine by weight |
Clinical Features of Hyperthyroidism
Symptoms:
- Weight loss despite increased appetite (↑ BMR)
- Palpitations, tachycardia, atrial fibrillation
- Heat intolerance, excessive sweating
- Diarrhoea, increased bowel frequency
- Tremor (fine, rest and intention)
- Anxiety, irritability, emotional lability, insomnia
- Menstrual irregularity, reduced fertility
- Muscle weakness (proximal myopathy)
- Dyspnoea (increased O₂ demand + AF → cardiac failure)
Signs specific to Graves disease:
- Exophthalmos (Proptosis): Caused by TSH receptor antibody stimulation of retro-orbital fibroblasts → glycosaminoglycan deposition → orbital volume expansion. Can occur WITHOUT hyperthyroidism. Ranged from mild proptosis to ophthalmoplegia and corneal exposure keratitis.
- Pretibial myxoedema: Glycosaminoglycan deposition in skin of lower legs — erythematous indurated plaques. Misnomer (it occurs in hyperthyroidism, not hypothyroidism)
- Thyroid acropachy: Clubbing with periosteal new bone formation (rare)
- Diffuse goitre: Bruit over thyroid (increased vascularity) — listen with stethoscope
Graves Disease — Detailed Pathophysiology
Graves disease is a type II hypersensitivity autoimmune disorder:
- Thyrotropin receptor antibodies (TRAb) — also called thyroid-stimulating immunoglobulins (TSI)
- These IgG autoantibodies bind and activate TSHR → mimic TSH → uncontrolled thyroid hormone production
- Additional antibodies: Anti-TPO (in ~75%), anti-thyroglobulin (in ~50%)
- Retro-orbital fibroblasts also express TSHR → TRAb stimulates these → Graves ophthalmopathy (independent of thyroid status)
- Trigger: Often stress, pregnancy, infection, iodine load
Investigations for Hyperthyroidism
| Test | Hyperthyroidism | Notes |
|---|---|---|
| TSH | ↓ (often undetectable) | Most sensitive screening test |
| Free T4 | ↑ | |
| Free T3 | ↑ | Sometimes T3 toxicosis with normal T4 |
| TRAb / TSI | Positive in Graves | Diagnostic |
| Anti-TPO | Positive in Graves/Hashimoto | |
| Radioactive iodine uptake (RAIU) | Diffuse ↑ in Graves; Focal in toxic adenoma; ↓ in thyroiditis | Distinguishes causes |
| Thyroid ultrasound | Diffuse hypervascularity in Graves | |
| Serum Tg | ↑ in all except factitious (exogenous T4) |
Treatment of Hyperthyroidism
Antithyroid drugs:
- Methimazole (carbimazole is pro-drug): Inhibits TPO → blocks organification and coupling. Standard first-line. Side effects: agranulocytosis (most dangerous — check WBC if fever/sore throat), rash, hepatotoxicity.
- Propylthiouracil (PTU): Inhibits TPO (same as methimazole) PLUS blocks peripheral T4→T3 conversion (5′-deiodinase inhibition). Preferred in: First trimester pregnancy (methimazole → aplasia cutis in foetus), thyroid storm. Higher risk of hepatotoxicity than methimazole (including fulminant hepatic failure → PTU only used when specifically indicated).
Beta-blockers (propranolol, atenolol):
- For symptom control — block adrenergic effects (tachycardia, tremor, anxiety)
- Propranolol at high doses also inhibits peripheral T4→T3 conversion
- Bridge therapy while antithyroid drugs take effect
Radioactive iodine (¹³¹I):
- Taken up by thyroid → emits β-radiation → destroys thyroid tissue
- Treatment of choice for Graves in many countries (not in pregnancy)
- Side effects: hypothyroidism (eventually in most), may worsen ophthalmopathy
- Contraindicated: pregnancy, active ophthalmopathy (relative), thyroid cancer (different protocol)
Surgery (thyroidectomy):
- Subtotal or total thyroidectomy
- Indicated: large goitre, malignancy suspected, thyroid storm refractory to medical treatment, patient preference
- Complications: Hypoparathyroidism (recurrent laryngeal nerve injury → hoarseness), hypothyroidism (subtotal)
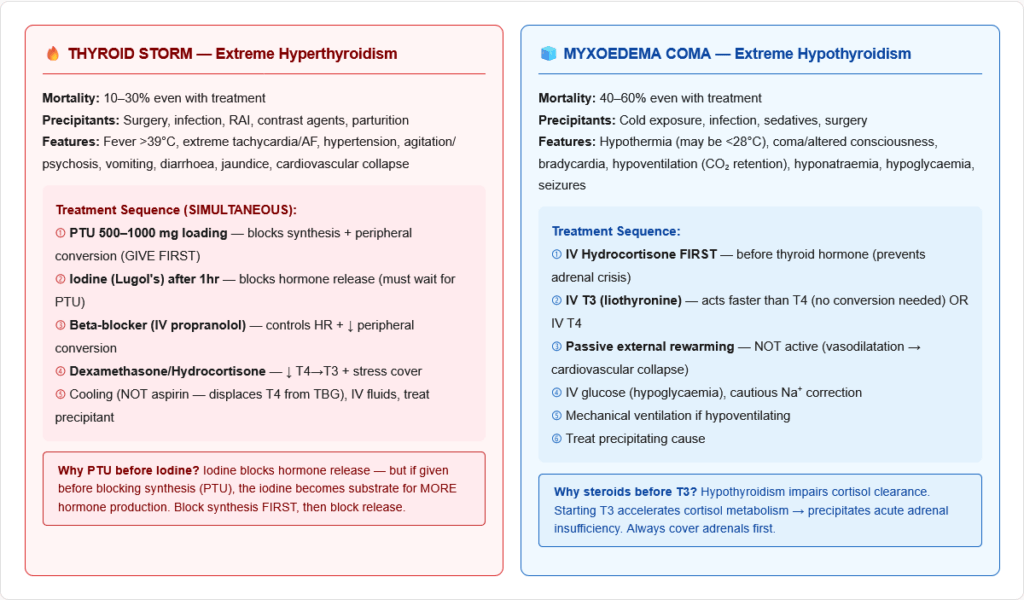
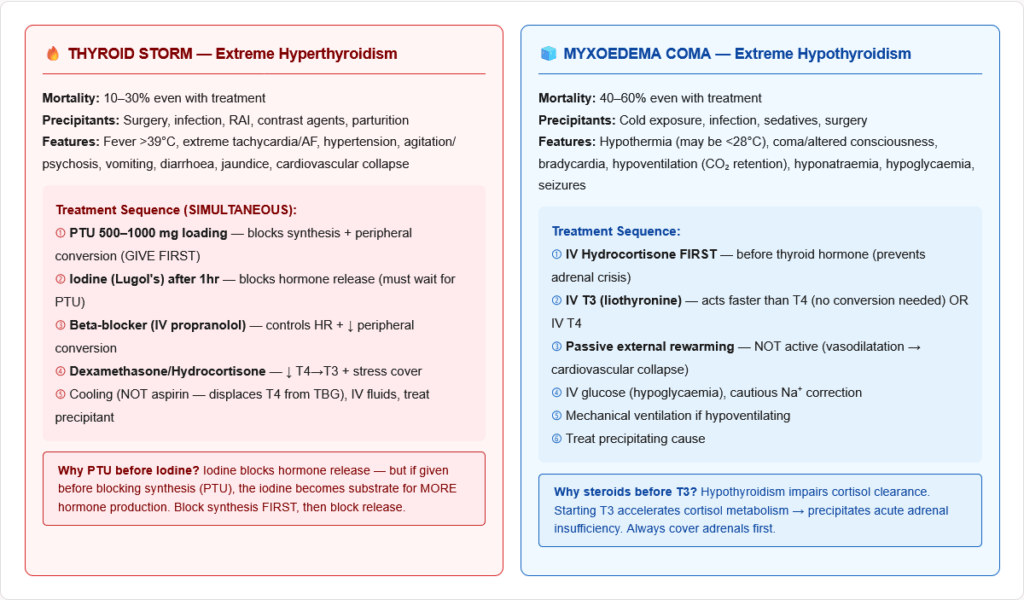
Thyroid Storm (Thyrotoxic Crisis)
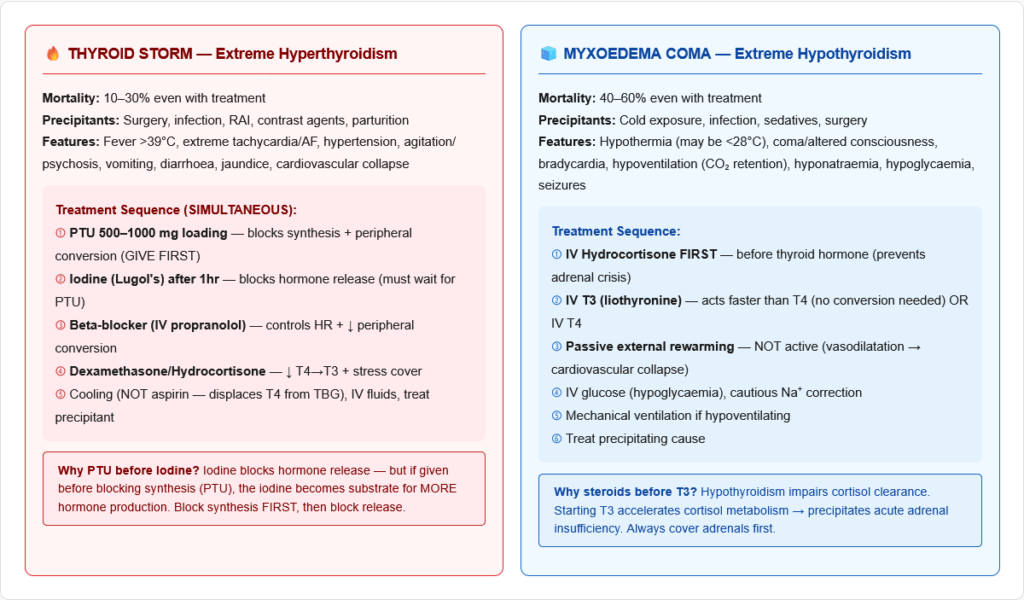
The most dangerous emergency in thyroid disease. Mortality 10–30% even with treatment.
Precipitants: Surgery, infection, trauma, radioactive iodine treatment, contrast agents, parturition, stopping antithyroid drugs
Features: High fever (>38.5°C, often >40°C), extreme tachycardia/AF, hypertension then cardiovascular collapse, severe agitation/psychosis/confusion, vomiting/diarrhoea, jaundice
Burch-Wartofsky Point Scale (BWPS): Clinical scoring system — score ≥45 = thyroid storm likely
Treatment (all simultaneously):
- PTU 500–1000 mg loading dose (blocks synthesis AND peripheral conversion) — given first
- Iodine (Lugol solution) — wait at least 1 hour after PTU (iodine blocks hormone release; but must block synthesis first or the iodine will be used to make MORE hormone)
- Beta-blocker (IV propranolol) — controls heart rate, reduces peripheral conversion
- Corticosteroids (IV dexamethasone/hydrocortisone) — inhibit peripheral T4→T3 conversion, provide stress cover, treat relative adrenal insufficiency
- Supportive: Cooling blankets (not aspirin — aspirin displaces T4 from TBG → worsens), IV fluids, treatment of precipitant
🏥 Section 7 — Hypothyroidism: Deficient Thyroid Hormone
Causes
| Type | Causes | Key Feature |
|---|---|---|
| Primary hypothyroidism (most common) | Hashimoto thyroiditis (#1 in iodine-sufficient countries), iodine deficiency (#1 worldwide), post-radioactive iodine, post-thyroidectomy, drugs (amiodarone, lithium), congenital | High TSH, low fT4 |
| Secondary (pituitary) | TSH-secreting tumour ablation, pituitary apoplexy, Sheehan syndrome | Low TSH, low fT4 |
| Tertiary (hypothalamic) | Craniopharyngioma, hypothalamic radiation | Low/normal TSH, low fT4 |
| Peripheral resistance | Thyroid hormone receptor mutation (RTH syndrome) | High TSH, high T4 — rare |
Hashimoto Thyroiditis — Detailed Pathophysiology
- Most common cause of hypothyroidism in iodine-sufficient areas
- Type IV hypersensitivity + humoral (type II) — cell-mediated + antibody-mediated autoimmune destruction
- Anti-TPO antibodies (most sensitive marker, >90% positive) and anti-thyroglobulin antibodies
- Histology: Diffuse lymphocytic infiltration with germinal centre formation, Hürthle cell (oxyphilic) change, follicular atrophy
- May present with transient hyperthyroidism (Hashitoxicosis) early in disease
- Associated with: HLA-DR3, HLA-DR5, other autoimmune diseases (Type 1 DM, Addison disease, vitiligo, pernicious anaemia, coeliac disease — APS type 2 / Schmidt syndrome)
- Increased risk of thyroid lymphoma (primary thyroid B-cell lymphoma — rare)
- Riedel thyroiditis: Rare variant — fibrous infiltration of thyroid and surrounding tissues → woody-hard thyroid → may compress trachea/oesophagus
Clinical Features of Hypothyroidism
Symptoms:
- Weight gain despite reduced appetite (↓ BMR)
- Cold intolerance
- Fatigue, lethargy, somnolence
- Constipation
- Dry skin, coarse hair, hair loss (especially outer third of eyebrows — Queen Anne’s sign)
- Hoarse voice (myxoedematous infiltration of vocal cords)
- Menorrhagia, infertility
- Depression, cognitive impairment (“myxoedema madness”)
- Carpal tunnel syndrome
- Muscle cramps, proximal myopathy
Signs:
- Bradycardia
- Delayed relaxation of tendon reflexes (most pathognomonic sign)
- Periorbital puffiness (non-pitting oedema from GAG deposition)
- Goitre (Hashimoto) or no goitre (post-thyroidectomy/radioiodine)
- Pericardial effusion
- Pleural effusion
- Myxoedema: Non-pitting oedema from deposition of glycosaminoglycans (GAGs — hyaluronic acid, chondroitin sulphate) in dermis and subcutaneous tissues. NOT the same as pitting oedema.
Investigations:
- TSH ↑ (primary — most sensitive test)
- Free T4 ↓
- Anti-TPO antibodies ↑ (Hashimoto)
- Cholesterol ↑ (LDL ↑ — reduced LDL receptor expression)
- CK ↑ (myopathy)
- Prolactin ↑ (TRH stimulates both TSH and prolactin → galactorrhoea)
- Anaemia (normocytic or macrocytic)
- Hyponatraemia (SIADH-like)
- ECG: Sinus bradycardia, low-voltage complexes, T-wave inversion
Treatment:
- Levothyroxine (L-T4, synthetic T4): Once-daily oral; peripherally converted to T3
- Starting dose: 1.6 µg/kg/day in young healthy adults; 25–50 µg/day in elderly/cardiac disease
- Monitor with TSH (target 0.5–2.5 mIU/L); allow 6 weeks between dose adjustments
- Take on empty stomach (absorption impaired by food, calcium, iron, cholestyramine, PPI)
Myxoedema Coma — The Extreme of Hypothyroidism
Medical emergency. Mortality ~40–60%.
Features: Hypothermia (may be severe — below 32°C), decreased consciousness/coma, bradycardia, hypoventilation (CO₂ retention), hyponatraemia, hypoglycaemia, seizures. Precipitated by infection, cold exposure, surgery, sedatives.
Treatment:
- IV T3 (liothyronine) or IV T4 (T3 acts faster)
- IV hydrocortisone (adrenal insufficiency may coexist — always treat before thyroid hormone)
- Passive external rewarming (not active — risk of vasodilatation and cardiovascular collapse)
- IV fluids with glucose, correct hyponatraemia cautiously
- Treat precipitating cause
- Mechanical ventilation if needed
Congenital Hypothyroidism (Cretinism)
Causes: Thyroid aplasia/dysplasia (most common), iodine deficiency, enzyme defects (e.g., TPO mutation, Pendred syndrome — TPO + deafness), ectopic thyroid
Features if untreated:
- Irreversible intellectual disability (most critical consequence)
- Deaf-mutism
- Short stature (epiphyseal dysgenesis)
- Coarse features, large tongue (macroglossia), umbilical hernia
- Pot-bellied, dry skin
- Hypothermia, poor feeding, prolonged jaundice in neonates
Newborn screening: Heel-prick blood spot test (TSH ± T4) at day 5–7 in UK, day 3–5 in USA. Identifies congenital hypothyroidism before symptoms develop.
Treatment: Start L-T4 as early as possible (within first 2 weeks of life) → if started early, intellectual development is completely normal.
🔬 Section 8 — Special Thyroid Conditions
Sick Euthyroid Syndrome (Non-Thyroidal Illness Syndrome, NTIS)
In any severe systemic illness (sepsis, MI, major surgery, starvation, burns), thyroid function tests become abnormal despite the thyroid being intrinsically normal.
Pattern:
- ↓ Total T3 (most consistent finding — reduced peripheral conversion)
- ↑ Reverse T3 (rT3) — alternative deiodination of T4
- Normal or ↓ T4 (in severe illness)
- Normal or ↓ TSH
Mechanism:
- ↓ Type I 5′-deiodinase (D1) activity → ↓ T4→T3 conversion
- ↑ Type III deiodinase (D3) → ↑ T4→rT3
- Inflammatory cytokines (IL-1, IL-6, TNF-α) directly suppress HPT axis
- Malnutrition reduces TBG → apparent ↓ total T4
Clinical importance:
- Do NOT treat — this is an adaptive response to severe illness (reduced metabolism = less energy expenditure = protective)
- The low T3 correlates with disease severity and prognosis
- Thyroid hormone replacement in sick euthyroid syndrome is harmful, not helpful
- TFTs should be rechecked 4–6 weeks after recovery
Subacute Granulomatous Thyroiditis (de Quervain Thyroiditis)
- Cause: Viral infection (mumps, Coxsackievirus, influenza, COVID-19)
- Pathology: Granulomatous inflammation of thyroid
- Clinical phases:
- Painful thyroid (tender, firm) + fever + elevated ESR/CRP
- Transient hyperthyroidism (2–6 weeks — destructive release of preformed hormones)
- Euthyroid phase
- Hypothyroid phase (may be permanent in 5–15%)
- Recovery to euthyroid
- Investigations: Low RAIU (destructive — gland not synthesising), elevated ESR, self-limiting
- Treatment: NSAIDs (pain), beta-blockers (hyperthyroid symptoms), corticosteroids (severe pain)
Postpartum Thyroiditis
- Autoimmune thyroiditis occurring within 1 year of delivery (similar to Hashimoto)
- Anti-TPO positive
- Affects 5–10% of women
- Same phases as subacute thyroiditis (hyperthyroid then hypothyroid)
- May recur with subsequent pregnancies
- Risk factor for permanent hypothyroidism and postnatal depression
Iodine Deficiency — A Global Problem
- Most common preventable cause of intellectual disability worldwide
- Dietary iodine <100 µg/day → insufficient substrate for T4 synthesis → ↑ TSH → thyroid hyperplasia → goitre
- Severe deficiency in pregnancy → endemic cretinism in offspring
- Prevention: Iodised salt (25–40 µg iodine/g salt) — one of the most cost-effective public health interventions
Drug Effects on Thyroid Function
| Drug | Effect on Thyroid | Mechanism |
|---|---|---|
| Amiodarone | Complex effects (see below) | 37% iodine by weight; D1/D2 inhibition |
| Lithium | Hypothyroidism ± goitre | Inhibits thyroid hormone release (blocks colloid resorption); inhibits T4→T3 |
| PTU | ↓ T3, ↓ T4 | TPO inhibition + D1 inhibition |
| Methimazole | ↓ T3, ↓ T4 | TPO inhibition only |
| High-dose iodine | Transient ↓ (Wolff-Chaikoff); if escapes → Jod-Basedow | |
| Phenytoin, carbamazepine | ↓ Total T4 | ↑ TBG catabolism; ↑ T4 metabolism |
| Glucocorticoids | ↓ TSH, ↓ T4→T3 | Suppress HPT axis; inhibit D1 |
| Aspirin/NSAIDs | ↓ Protein binding | Displace T4 from TBG |
| Dopamine | ↓ TSH | Inhibits pituitary thyrotrophs |
| Oestrogens (OCP/pregnancy) | ↑ TBG → ↑ total T4 | Free T4 normal |
Amiodarone and Thyroid Disease (Special Mention)
Amiodarone is a unique and complex drug regarding thyroid effects:
- Contains 37% iodine by weight → massive iodine load
- Has very long half-life (~100 days) → effects persist for months after stopping
- Always causes: ↑ T4, ↓ T3, ↓ TSH initially (due to D1 inhibition — reduces peripheral T4→T3 conversion)
- Can cause either hypothyroidism OR hyperthyroidism:
Amiodarone-induced Hypothyroidism (AIH):
- Due to Wolff-Chaikoff effect — excess iodine blocks organification
- More common in iodine-sufficient areas and patients with underlying Hashimoto
- Treatment: Levothyroxine; continue amiodarone if needed for arrhythmia
Amiodarone-induced Thyrotoxicosis (AIT):
- Type I: Excess iodine → autonomous hormone synthesis (in underlying nodular disease); treat with antithyroid drugs
- Type II: Destructive thyroiditis → release of preformed hormones; treat with glucocorticoids
- Mixed type: Combined — often requires combined treatment
- KClO₄ (perchlorate) can be added to block NIS in Type I AIT
🔬 Section 9 — Thyroid Cancer
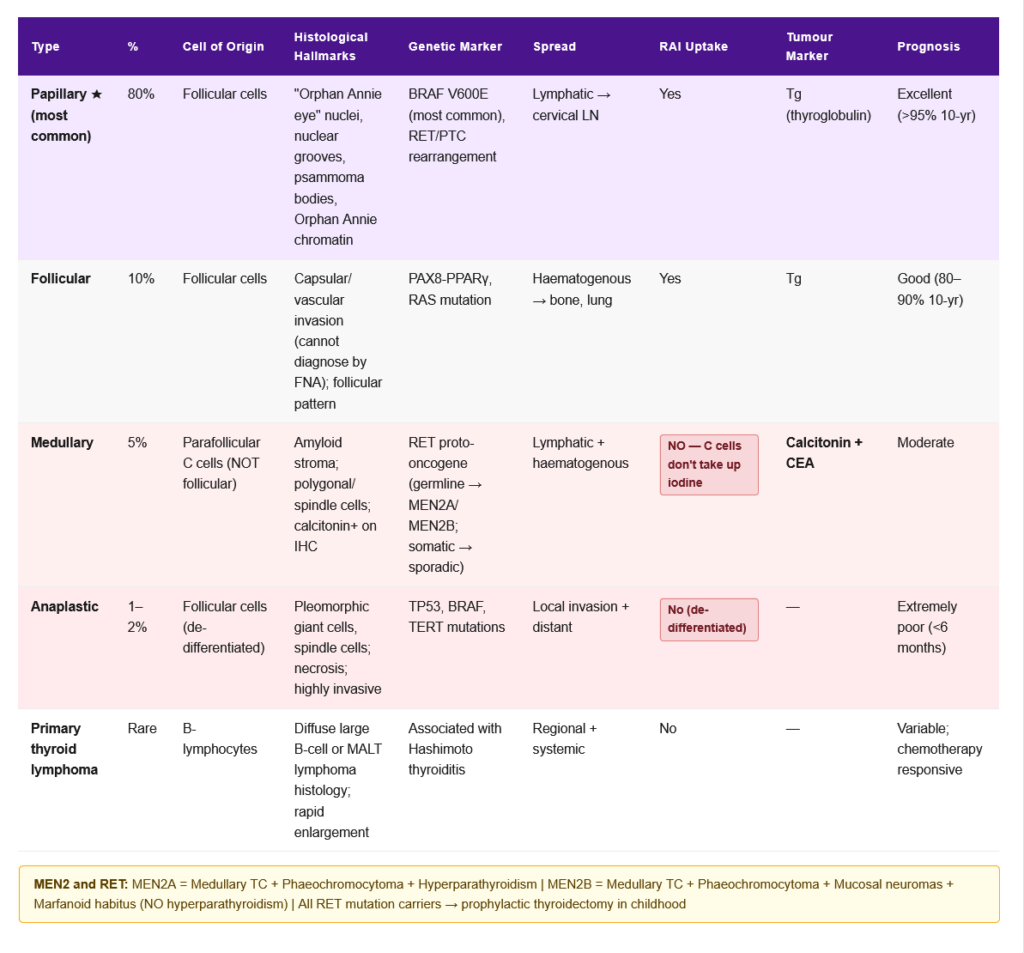
Classification and Features
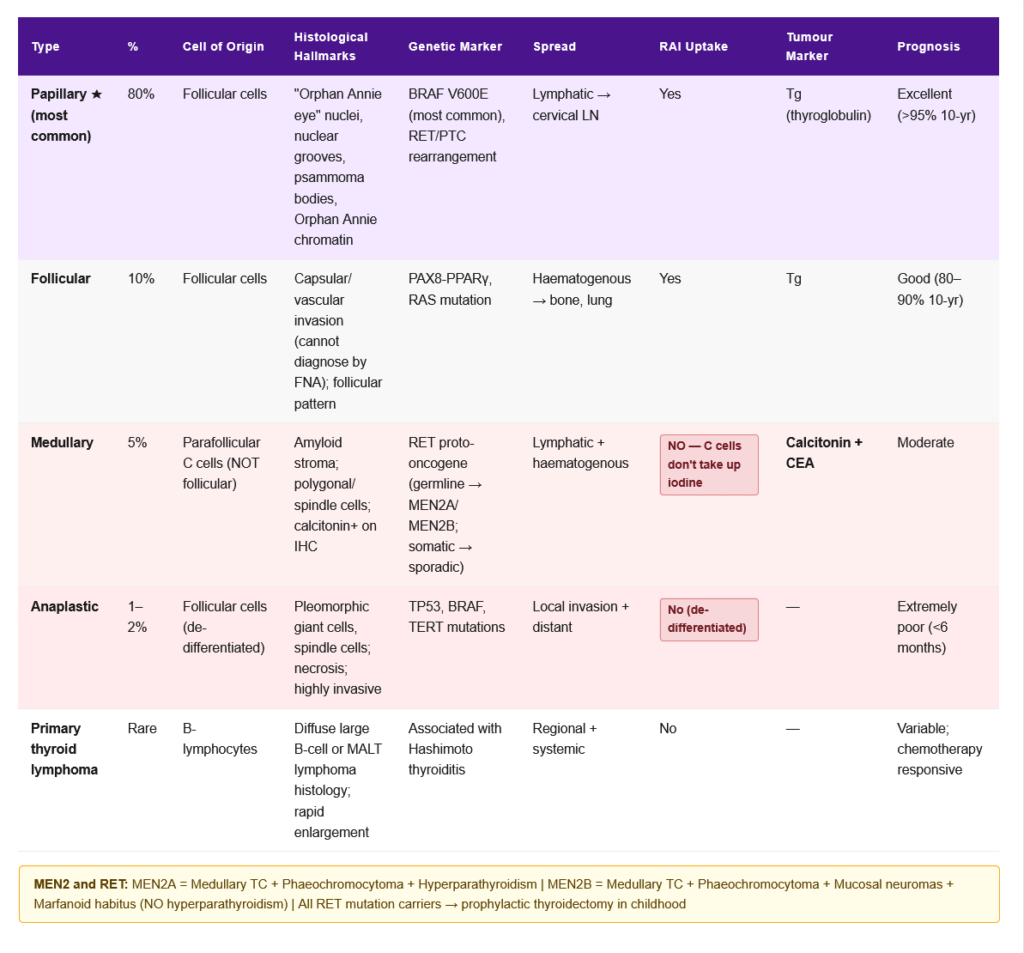
| Type | Frequency | Origin | Key Features | Treatment | Prognosis |
|---|---|---|---|---|---|
| Papillary | ~80% | Follicular cells | “Orphan Annie eye” nuclei, psammoma bodies, nuclear grooves; RET/PTC rearrangement; BRAF V600E mutation; slow-growing; lymph node mets | Surgery + RAI + TSH suppression | Excellent (>95% 10-yr survival) |
| Follicular | ~10% | Follicular cells | Capsular/vascular invasion (FNA cannot diagnose); PAX8-PPARγ, RAS mutations; haematogenous spread (bone, lung) | Surgery + RAI | Good (80–90% 10-yr) |
| Medullary | ~5% | Parafollicular C cells | Secretes calcitonin (tumour marker); amyloid stroma; MEN2A/MEN2B (RET mutation); calcitonin-CEA useful markers | Surgery (total thyroidectomy); RAI NOT effective | Moderate |
| Anaplastic | ~1–2% | Follicular cells (de-differentiated) | Most aggressive cancer; tracheal invasion; rapidly fatal | Palliative | Extremely poor (<6 months) |
| Primary thyroid lymphoma | Rare | B-lymphocytes | Associated with Hashimoto; rapid thyroid enlargement | Chemotherapy + radiation | Variable |
RET Proto-oncogene:
- Germline mutations in RET → familial medullary thyroid carcinoma (FMTC) and MEN2
- MEN2A: Medullary thyroid cancer + phaeochromocytoma + primary hyperparathyroidism
- MEN2B: Medullary thyroid cancer + phaeochromocytoma + marfanoid habitus + mucosal neuromas (NO hyperparathyroidism)
- All RET mutation carriers should undergo prophylactic thyroidectomy in childhood
🔄 Section 10 — Connections to Other Systems
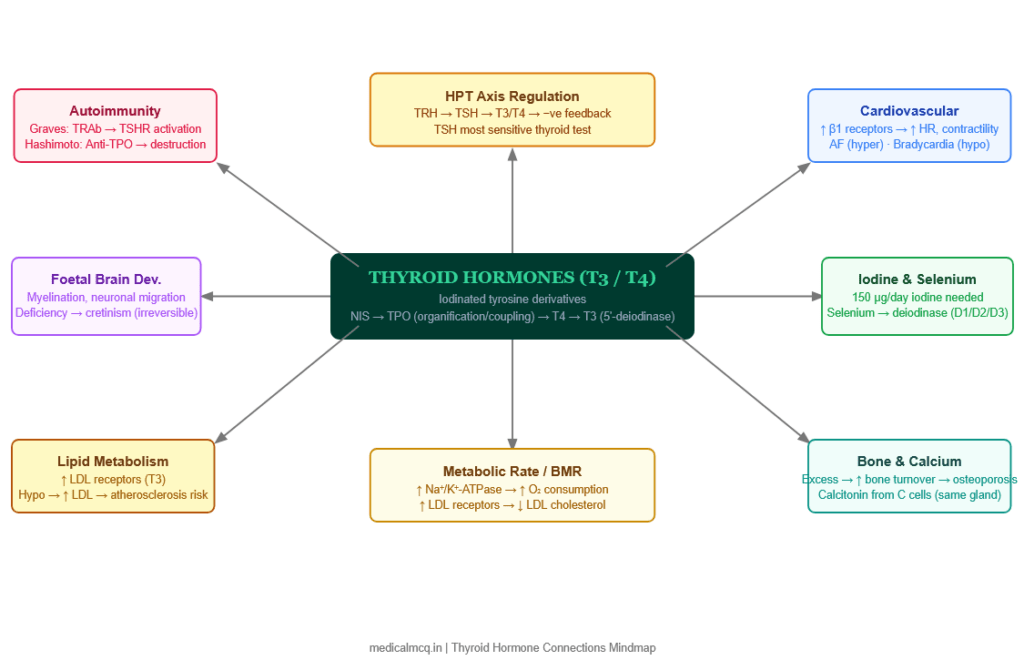
→ Cholesterol and Lipid Metabolism — T3 upregulates LDL receptors → ↑ LDL clearance from blood. Hypothyroidism → ↓ LDL receptors → ↑ LDL → dyslipidaemia → premature atherosclerosis. This is one of the most important clinical consequences of subclinical hypothyroidism.
→ Steroid Hormone Synthesis — Cortisol suppresses TSH secretion and inhibits peripheral T4→T3 conversion. TRH stimulates both TSH and prolactin (relevant in hypothyroidism → galactorrhoea from elevated TRH → ↑ prolactin). Oestrogen ↑ TBG → apparent ↑ total T4 in pregnancy/OCP.
→ Calcium Metabolism — Calcitonin is secreted by parafollicular C cells in the thyroid (same organ, different cell type). Medullary thyroid cancer secretes excess calcitonin. Hyperthyroidism → ↑ bone turnover → hypercalciuria (mild hypercalcaemia occasionally). Post-thyroidectomy → hypoparathyroidism risk.
→ Iron and Haematopoiesis — Thyroid hormones stimulate EPO production → normal erythropoiesis. Hypothyroidism → ↓ EPO → normocytic anaemia. Autoimmune hypothyroidism (Hashimoto) often coexists with pernicious anaemia (autoimmune) → macrocytic anaemia + B12 deficiency.
→ Iodine and Nutrition — Dietary iodine requirement: 150 µg/day (adults); 220 µg/day (pregnancy); 290 µg/day (lactation). Selenium is needed for deiodinase enzymes (D1, D2, D3 are selenoproteins). Selenium deficiency → impaired T4→T3 conversion → hypothyroid-like state.
→ Foetal Development — Maternal T4 (crosses placenta before foetal thyroid develops at 12 weeks) is essential for foetal brain development. Foetal D3 in placenta protects foetus from excess maternal T4. MCT8 transporter is essential for T3 entry into brain cells (mutations → Allan-Herndon-Dudley syndrome).
🎯 High-Yield Exam Facts
🔴 TSH is the single most sensitive and specific test for thyroid function in primary thyroid disease Low TSH = hyperthyroidism; High TSH = primary hypothyroidism. The log-linear relationship means small changes in free T4 cause large changes in TSH. Always use TSH as first-line test; add free T4 if TSH is abnormal.
🔴 PTU vs Methimazole: PTU inhibits BOTH TPO AND peripheral T4→T3 conversion; Methimazole inhibits TPO only PTU is preferred in: (1) Thyroid storm, (2) First trimester pregnancy. Methimazole is preferred at all other times (more convenient dosing, less hepatotoxicity).
🔴 Graves disease = TSH receptor antibodies (TRAb/TSI) stimulate TSHR → uncontrolled T4/T3 production The ophthalmopathy is caused by TRAb acting on orbital fibroblasts — it can be independent of thyroid function. Anti-TPO antibodies are also positive in ~75% of Graves.
🔴 Congenital hypothyroidism: Newborn screening (day 3–7 TSH) + early L-T4 treatment = normal intellectual development Without treatment → irreversible cretinism. This is why newborn screening is one of the most important public health programmes in medicine.
🔴 Iodine excess causes Wolff-Chaikoff effect (blocks TPO) — protective; but Jod-Basedow in susceptible patients The thyroid normally “escapes” from Wolff-Chaikoff after 1–2 weeks. Amiodarone (37% iodine) disrupts this escape mechanism in diseased thyroids.
🔴 Sick euthyroid syndrome: Low T3, high rT3, normal/low TSH — Do NOT treat with thyroid hormones This is an adaptive response to severe illness. Treatment with T3/T4 is harmful. rT3 is the key distinguishing marker from true hypothyroidism (rT3 is HIGH in sick euthyroid, but LOW or normal in true hypothyroidism).
🟠 Oestrogen/OCP/Pregnancy → ↑ TBG → ↑ total T4 but FREE T4 and TSH are NORMAL This is a TBG change, NOT hyperthyroidism. Same principle as cortisol and CBG. Always measure FREE hormones to assess true status.
🟠 Medullary thyroid cancer: calcitonin is the tumour marker; arises from C cells; associated with RET mutation and MEN2 Calcitonin is the diagnostic and monitoring marker. MEN2A = MTC + phaeochromocytoma + hyperparathyroidism. MEN2B = MTC + phaeochromocytoma + mucosal neuromas + marfanoid habitus.
🟠 Amiodarone: 37% iodine; inhibits D1 → ↑ T4, ↓ T3 on baseline TFTs; can cause EITHER hypothyroidism OR hyperthyroidism Always monitor TFTs every 6 months in patients on amiodarone. Type I AIT = antithyroid drugs; Type II AIT = glucocorticoids.
🟠 Lithium causes hypothyroidism by blocking thyroid hormone secretion (colloid resorption) Monitor TFTs in all patients on long-term lithium. Also inhibits peripheral T4→T3 conversion.
🟡 Thyroid storm treatment: PTU → then Iodine → Beta-blocker + Steroids + Supportive The sequence matters: PTU must be given at least 1 hour before iodine — iodine blocks hormone release but if given first, the excess iodine will be used to synthesise more hormone. “Block first, then release.”
🟡 In myxoedema coma, give corticosteroids BEFORE thyroid hormone Hypothyroidism often coexists with relative adrenal insufficiency. Starting T3/T4 before steroid cover can precipitate adrenal crisis by increasing cortisol metabolism.
🟡 Papillary thyroid cancer: most common thyroid cancer (80%); “Orphan Annie eye” nuclei; psammoma bodies; BRAF V600E Spread is lymphatic (cervical lymph nodes). Haematogenous spread (bone, lung) is more typical of follicular carcinoma.
🟡 Hashimoto thyroiditis: anti-TPO antibodies; diffuse lymphocytic infiltration; Hürthle cell change; increased risk of thyroid lymphoma Anti-TPO is the most sensitive antibody marker. Association with HLA-DR3/DR5 and other autoimmune diseases (Type 1 DM, Addison, vitiligo, pernicious anaemia).
🧠 Mnemonics & Memory Tricks
“MIG COUPLING” — Steps of Thyroid Hormone Synthesis → Helps remember the complete synthesis sequence
M = MIT and DIT formed (monoiodo, diiodo) I = Iodide trapped by NIS (sodium-iodide symporter) G = Gland (thyroglobulin) stores T3/T4 in colloid COUPLING = MIT + DIT → T3; DIT + DIT → T4 (by TPO) I = Iodide recycled from MIT/DIT by dehalogenase N = NaI symporter G = GTP-coupled TSHR (Gαs → cAMP) mediates ACTH-like stimulation
💡 Simplified sequence to memorise: Iodide Trap → Oxidise → Organify → Couple → Store → Resorb → Proteolysis → Secrete → Peripheral convert (T4→T3)
“TPO POiSON” — What Inhibits Thyroid Peroxidase (TPO) T = Thiourea drugs (PTU, Methimazole, Carbimazole) P = PTU also blocks Peripheral conversion (extra action) O = Organification is blocked P = Perchlorate blocks NIS (not TPO, but related) O = Only PTU used in thyroid storm + first trimester pregnancy I = Iodine excess (Wolff-Chaikoff) — transient S = Storm → use PTU not methimazole O = Other: PTU in breastfeeding preferred (less transfer) N = Never use methimazole in first trimester (aplasia cutis)
💡 Pro tip: Methimazole = “MOST of the time.” PTU = “Particular situations” (Storm, Pregnancy 1st trimester).
“GRAVES” — Features of Graves Disease G = Goitre (diffuse, with bruit) R = Re-orbital disease (exophthalmos/proptosis) A = Anti-TSH receptor antibodies (TRAb/TSI) V = Very high T4, very low TSH E = Eye disease (ophthalmopathy — can occur independently) S = Skin — pretibial myxoedema + acropachy
💡 Pro tip: The ophthalmopathy of Graves is caused by TRAb acting on ORBITAL fibroblasts — it is NOT caused by thyroid hormones. That is why treating the thyroid doesn’t always fix the eyes.
“5Ds of Cretinism” — Features of Untreated Congenital Hypothyroidism D = Dull (intellectual disability — most critical) D = Deaf-mute D = Dwarf (short stature — epiphyseal dysgenesis) D = Dry skin, coarse features, macroglossia D = Delayed bone age
💡 Pro tip: All 5Ds are preventable with early L-T4 treatment started within 2 weeks of birth. Newborn screening (TSH on day 3–7) exists precisely to prevent this.
⚠️ Common Mistakes Students Make on This Topic
❌ Mistake: “TSH is low in ALL types of hypothyroidism” ✅ Reality: TSH is HIGH in PRIMARY hypothyroidism (the pituitary increases TSH in response to inadequate thyroid hormone). TSH is LOW or inappropriately normal in SECONDARY (pituitary) or TERTIARY (hypothalamic) hypothyroidism. Always determine whether TSH is high or low BEFORE concluding the cause. 📝 How this gets tested: “A patient has low T4 and low TSH — what is the most likely cause?” → Secondary (pituitary) hypothyroidism, not primary. Low TSH + low T4 = look at the pituitary, not the thyroid.
❌ Mistake: “Pretibial myxoedema occurs in hypothyroidism” ✅ Reality: Despite its misleading name, pretibial myxoedema occurs in HYPERTHYROIDISM — specifically Graves disease. It is caused by TSH receptor antibody stimulation of dermal fibroblasts → glycosaminoglycan deposition in skin of lower legs → indurated, erythematous plaques. The word “myxoedema” in this context refers to the local skin change, not the systemic hypothyroid state. 📝 How this gets tested: “A patient with hyperthyroidism has indurated plaques on the shins — diagnosis?” → Pretibial myxoedema of Graves disease.
❌ Mistake: “In sick euthyroid syndrome, the patient needs thyroid hormone replacement” ✅ Reality: Sick euthyroid syndrome is an adaptive response — the body deliberately reduces T3 to lower metabolic rate during critical illness. Treating with thyroid hormones is harmful. The low T3 level correlates with disease severity but is NOT a treatment target. TFTs should be rechecked after full recovery. 📝 How this gets tested: “An ICU patient with sepsis has low T3, elevated rT3, normal TSH — management?” → Supportive care for the underlying illness; do NOT start levothyroxine.
❌ Mistake: “PTU and methimazole have the same mechanism and indications” ✅ Reality: Both inhibit TPO (organification/coupling), but PTU ALSO inhibits 5′-deiodinase Type I (peripheral T4→T3 conversion) — methimazole does not. This extra action makes PTU the drug of choice in thyroid storm (needs rapid reduction of both synthesis AND circulating T3). Methimazole is preferred in all other situations (once-daily dosing, less hepatotoxicity). Methimazole is CONTRAINDICATED in first trimester (aplasia cutis congenita in foetus). 📝 How this gets tested: “Which antithyroid drug inhibits peripheral conversion of T4 to T3?” = PTU. “Which antithyroid drug is preferred in thyroid storm?” = PTU. “Which is preferred for long-term management of Graves in a non-pregnant adult?” = Methimazole.
❌ Mistake: “Radioactive iodine (¹³¹I) can be used to image medullary thyroid cancer” ✅ Reality: Medullary thyroid cancer arises from parafollicular C cells, which do NOT take up iodine (they don’t make thyroid hormones — they make calcitonin). ¹³¹I scanning and ablation are ONLY useful for differentiated thyroid cancers (papillary and follicular) that arise from follicular cells and retain the NIS transporter. Medullary cancer is imaged with MIBG scan or PET-CT (if dedifferentiated). 📝 How this gets tested: “A patient with medullary thyroid cancer undergoes ¹³¹I ablation — what is expected?” → No uptake; the treatment is ineffective. Calcitonin is the relevant marker for medullary cancer.
📝 5 Practice MCQs — Test Yourself Now
Q1: A 32-year-old woman presents with 3 months of palpitations, weight loss, heat intolerance, and a fine tremor. TSH is 0.01 mIU/L and free T4 is 42 pmol/L. TSH receptor antibodies are positive. Which drug combination should be used if she is found to be 8 weeks pregnant?
- A. Methimazole + propranolol
- B. PTU + propranolol
- C. Radioactive iodine + atenolol
- D. Methimazole + corticosteroids
✅ Answer: B. PTU + propranolol
Why correct: This patient has Graves disease (positive TRAb, clinical features of hyperthyroidism). She is in the first trimester (8 weeks). The critical choice here is PTU over methimazole in the first trimester — methimazole is associated with aplasia cutis congenita (absent skin patch on scalp) and the methimazole embryopathy (choanal atresia, oesophageal atresia) when used in the first trimester. PTU does not carry this teratogenic risk in the first trimester. After the first trimester (12–14 weeks), it is usual to switch to methimazole (because PTU carries higher risk of maternal hepatotoxicity). Propranolol is used for symptomatic relief of tachycardia and tremor — it is safe in pregnancy at low doses for short-term use.
Why A is wrong: Methimazole is CONTRAINDICATED in the first trimester because of aplasia cutis and methimazole embryopathy. Why C is wrong: Radioactive iodine (¹³¹I) is absolutely CONTRAINDICATED in pregnancy — it crosses the placenta and would ablate the foetal thyroid (which begins iodide uptake from ~12 weeks). This would cause congenital hypothyroidism and cretinism. Why D is wrong: Methimazole is contraindicated in first trimester. Corticosteroids have no primary role in treating hyperthyroidism of Graves disease (they are used in thyroid storm as an adjunct).
Exam tip: Pregnancy + hyperthyroidism + first trimester = PTU. Pregnancy + hyperthyroidism + second/third trimester = methimazole (lower PTU hepatotoxicity risk). Radioactive iodine = NEVER in pregnancy.
Q2: A 55-year-old woman with known Hashimoto thyroiditis is started on high-dose amiodarone for refractory ventricular tachycardia. 6 months later, her TFTs show: TSH <0.01 mIU/L, free T4 markedly elevated, free T3 elevated. Radioactive iodine uptake scan shows very LOW uptake. Which type of amiodarone-induced thyrotoxicosis is this, and what is the correct treatment?
- A. Type I AIT (excess iodine-driven synthesis); treat with carbimazole
- B. Type II AIT (destructive thyroiditis); treat with prednisolone
- C. Graves disease triggered by amiodarone; treat with radioactive iodine
- D. Factitious thyrotoxicosis from amiodarone iodine; observe and recheck in 6 months
✅ Answer: B. Type II AIT (destructive thyroiditis); treat with prednisolone
Why correct: The key distinguishing feature between Type I and Type II AIT is the radioactive iodine uptake (RAIU). In Type II AIT (destructive thyroiditis), the thyroid gland is damaged by amiodarone and inflammatory mediators → preformed hormones are released passively → the gland is NOT actively synthesising hormones → RAIU is very low. In Type I AIT (excess iodine drives autonomous hormone synthesis in nodular disease), the gland is actively making hormone → RAIU is normal or high. Additionally, this patient has Hashimoto thyroiditis — an underlying autoimmune thyroid condition — which is a risk factor for amiodarone-induced destructive thyroiditis. Treatment of Type II AIT is glucocorticoids (prednisolone) which suppress the inflammatory/destructive process.
Why A is wrong: Type I AIT has NORMAL or HIGH radioactive iodine uptake (active synthesis). Treatment of Type I is antithyroid drugs (carbimazole/PTU) ± potassium perchlorate to block NIS. Why C is wrong: Graves disease would show DIFFUSELY HIGH radioactive iodine uptake with positive TRAb. Also, radioactive iodine treatment is avoided in amiodarone-induced thyrotoxicosis because the gland is already loaded with iodine (amiodarone’s iodine saturates the gland → RAI uptake would be poor anyway). Why D is wrong: Factitious thyrotoxicosis would show very low thyroglobulin (exogenous hormone — no thyroid stimulation). This patient is clinically thyrotoxic with elevated T4 and T3 — observation is inappropriate; treatment is needed.
Exam tip: AIT Type I = low-uptake area + underlying nodular disease → antithyroid drugs. AIT Type II = low-uptake + underlying normal/Hashimoto thyroid → steroids. The RAIU result is the pivot of this question.
Q3: A 45-year-old man with severe septic shock is admitted to the ICU. His TFTs show: TSH 0.8 mIU/L (normal), free T4 9 pmol/L (low), free T3 1.2 pmol/L (very low), reverse T3 markedly elevated. What is the most appropriate next step?
- A. Start IV liothyronine (T3) immediately — the patient has hypothyroidism
- B. Start levothyroxine (T4) 100 µg daily — to correct the low T4
- C. Treat the underlying sepsis; the TFT abnormalities represent sick euthyroid syndrome and do not require thyroid hormone replacement
- D. Measure anti-TPO antibodies and TSH receptor antibodies to determine the aetiology
✅ Answer: C. Treat the underlying sepsis; the TFT abnormalities represent sick euthyroid syndrome and do not require thyroid hormone replacement
Why correct: The clinical picture is classic sick euthyroid syndrome (NTIS): low free T3, elevated reverse T3, relatively normal TSH, in the context of severe acute illness (septic shock). The mechanism is cytokine-mediated suppression of D1 (reducing T4→T3 conversion) and upregulation of D3 (increasing T4→rT3). This is an adaptive response to reduce metabolic rate and preserve energy during critical illness. The normal TSH rules out primary or secondary hypothyroidism. Multiple randomised trials have shown that thyroid hormone supplementation in sick euthyroid syndrome does NOT improve outcomes and may cause harm.
Why A is wrong: IV T3 is indicated for myxoedema coma (where the patient has TRUE hypothyroidism with very high TSH, cold temperature, altered consciousness from chronic hypothyroid state). This patient’s TSH is normal — this excludes true primary hypothyroidism. Why B is wrong: Levothyroxine supplementation in sick euthyroid syndrome is not indicated and has been shown to not improve and potentially worsen outcomes. The low T4 is a consequence of severe illness, not a primary thyroid problem. Why D is wrong: While antibody testing can be part of a workup if hypothyroidism is suspected after recovery, it is not the immediate management priority in an ICU patient with sick euthyroid syndrome. TFTs should be rechecked 4–6 weeks after recovery from the acute illness.
Exam tip: The distinguishing feature of sick euthyroid from true hypothyroidism: sick euthyroid has ELEVATED rT3 + NORMAL TSH. True primary hypothyroidism has HIGH TSH + LOW T4 + LOW or NORMAL rT3. The rT3 and TSH are the two most useful distinguishing tests.
Q4: A 6-week-old infant is referred after a positive newborn heel-prick screen. TSH is 280 mIU/L, free T4 is undetectable. The baby appears normal on examination. Thyroid ultrasound shows no thyroid tissue in the normal position but identifies ectopic thyroid tissue at the base of the tongue. What is the most important aspect of management?
- A. Reassure the parents and repeat TFTs at 6 months
- B. Surgically remove the ectopic thyroid tissue as soon as possible
- C. Start oral levothyroxine immediately without waiting for further confirmatory tests
- D. Measure anti-TPO antibodies; if positive, start treatment only then
✅ Answer: C. Start oral levothyroxine immediately without waiting for further confirmatory tests
Why correct: This infant has congenital hypothyroidism from thyroid ectopia (lingual thyroid — most common ectopic location). The TSH of 280 mIU/L with undetectable T4 confirms severe primary hypothyroidism. The most critical management priority is to start L-T4 immediately — ideally within the first 2 weeks of life. The brain is exquisitely dependent on thyroid hormones for development in the first 2–3 years. Every day of untreated hypothyroidism risks permanent intellectual disability. When started early, treated congenital hypothyroid infants have normal intellectual development. Waiting for additional tests or antibodies is not justified — the biochemical diagnosis is clear.
Why A is wrong: Reassurance and waiting is dangerous. Untreated congenital hypothyroidism causes irreversible intellectual disability (cretinism). The window for effective treatment is narrow — treatment must begin within weeks of birth. Why B is wrong: The ectopic thyroid tissue (lingual thyroid) should NOT be removed — it may be the only functioning thyroid tissue the infant has. Removing it would worsen the hypothyroidism. It should be left in place (the infant is already on L-T4 replacement). Why D is wrong: Anti-TPO antibodies are relevant for diagnosing Hashimoto thyroiditis — not thyroid ectopia. Waiting for antibody results would dangerously delay treatment. The biochemical picture of TSH 280 + undetectable T4 + ultrasound confirming ectopic thyroid is already diagnostic and sufficient to begin treatment.
Exam tip: Congenital hypothyroidism = treat immediately with L-T4 once diagnosis is biochemically confirmed. Do not wait. The intellectual outcome is directly related to how early treatment begins. Target TSH should be normalised within 2–4 weeks of starting treatment.
Q5: A 70-year-old woman presents with acute confusion, hypothermia (32°C), bradycardia (42 bpm), and facial puffiness. Her husband says she has been on no medications. Random cortisol is 150 nmol/L (reference >550 nmol/L in acute stress). TSH is 85 mIU/L, free T4 is undetectable. What is the correct sequence of treatment?
- A. IV T4 alone → then add corticosteroids if no improvement at 24 hours
- B. IV corticosteroids first → then IV T3 (or T4) + rewarming + supportive care
- C. Oral levothyroxine + active external rewarming + IV thiamine
- D. IV T3 alone — the low cortisol is incidental and does not require treatment
✅ Answer: B. IV corticosteroids first → then IV T3 (or T4) + rewarming + supportive care
Why correct: This is myxoedema coma (altered consciousness + hypothermia + bradycardia + very high TSH + undetectable T4 in an elderly woman). The critical and frequently tested management point is: Give corticosteroids BEFORE thyroid hormone. The reason: hypothyroidism impairs cortisol metabolism, and cortisol secretion may be borderline even at rest. When thyroid hormone is given, it rapidly accelerates cortisol metabolism → can precipitate acute adrenal crisis. This patient already has an inappropriately low cortisol (150 nmol/L) during an acute stress — this confirms relative adrenal insufficiency and makes steroid cover even more urgent. After corticosteroids: IV T3 (liothyronine) acts faster than T4 (no peripheral conversion required) and is preferred in coma. Passive external rewarming only (active rewarming causes peripheral vasodilation and cardiovascular collapse).
Why A is wrong: Starting IV T4 without steroid cover risks precipitating adrenal crisis. In myxoedema coma, adrenal insufficiency must always be covered before thyroid hormone is given. Why C is wrong: Oral levothyroxine is absorbed unreliably in myxoedema coma (reduced GI motility, impaired absorption) — IV T3 or T4 is preferred. Active external rewarming (e.g., warming blankets) is contraindicated — it causes peripheral vasodilation and vascular collapse. Why D is wrong: IV T3 alone without steroid cover is dangerous (adrenal crisis). The low cortisol in acute stress is NOT incidental — it must be treated. Always give steroid cover first.
Exam tip: Myxoedema coma management sequence: ① Steroid (hydrocortisone IV) first ② Then IV T3/T4 ③ Passive rewarming (NOT active) ④ Treat precipitant ⑤ IV glucose ⑥ Mechanical ventilation if needed. The steroid-first rule is the most testable fact about myxoedema coma treatment.
📚 References
📖 Harrison’s Principles of Internal Medicine — Kasper et al. | Chapter 375: Disorders of the Thyroid Gland
📖 Williams Textbook of Endocrinology — Melmed et al. | Chapter 11: Thyroid Physiology and Diagnostic Evaluation; Chapter 12: Hypothyroidism and Thyroiditis; Chapter 13: Hyperthyroidism
📖 Guyton and Hall Textbook of Medical Physiology — Hall | Chapter 76: Thyroid Metabolic Hormones
📖 Harper’s Illustrated Biochemistry — Rodwell et al. | Chapter 41: Hormones of the Thyroid Gland
📖 Katzung’s Basic & Clinical Pharmacology — Katzung | Chapter 38: Thyroid and Antithyroid Drugs
📖 Robbins Basic Pathology — Kumar et al. | Chapter 20: The Endocrine System — Thyroid
🚀 Keep Practising — You Are Not Done Yet
Thyroid questions in NEET PG and USMLE span biochemistry (synthesis pathway, enzyme defects), physiology (HPT axis, feedback), pharmacology (PTU vs methimazole, amiodarone effects), and full clinical endocrinology (Graves, Hashimoto, storm, coma, cancer). No other endocrine topic spans this many disciplines simultaneously.
The students who score highest are those who understand each condition mechanistically — they can explain WHY the tremor occurs in Graves, WHY the reflex relaxation is delayed in hypothyroidism, WHY PTU not methimazole in first trimester, WHY steroids before T3 in myxoedema coma. That level of understanding only comes from structured practice with clinical-scenario MCQs.
medicalmcq.in has free Biochemistry and Endocrinology MCQs covering every topic in this article — all in clinical-scenario format with detailed mechanistic explanations.