What You Will Learn in This Article
- Why phenylalanine is classified as a conditionally essential amino acid precursor of tyrosine
- The enzyme, cofactor, and mechanism of the phenylalanine → tyrosine conversion
- The complete downstream metabolic map of tyrosine — what it becomes and where
- Classic genetic disorders arising from enzyme defects along this pathway
- Clinical presentations, lab findings, and dietary management for PKU and other disorders
- High-yield facts that are repeatedly tested across NEET PG, USMLE, AIIMS, and FMGE
- Mnemonics to lock in the pathway permanently
📖 Introduction: Why This Topic Matters in Exams
Imagine a newborn who seems perfectly healthy at birth. Over the first few months of life, developmental milestones slip. By age 1, intellectual disability is becoming apparent. The parents notice a musty, “mousy” odour from the child’s urine. A heel-prick screening test done at birth had flagged an abnormal result — but the family was never followed up.
This is the story of untreated Phenylketonuria (PKU) — and it begins with one enzyme failing to convert phenylalanine into tyrosine.
This topic is a perennial favourite in medical entrance examinations for several reasons:
- It connects biochemistry directly to clinical medicine — textbook enzyme names translate directly into real diseases with recognizable presentations.
- It requires integrated thinking — one pathway connects amino acid biochemistry, neurotransmitter synthesis, pigment biology, thyroid hormone metabolism, and neonatal screening.
- It is rich in testable eponyms, enzyme names, and classic “spot diagnosis” clinical vignettes.
Boards love this topic because it punishes candidates who memorise in isolation. Read this article and you will own the whole map.
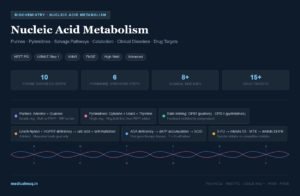
🔬 Section 1 — The Phenylalanine to Tyrosine Conversion: Foundational Biochemistry
1A. The Core Reaction
Phenylalanine is an essential amino acid — the body cannot synthesise it and must obtain it from dietary protein (meat, dairy, legumes). Tyrosine, on the other hand, is classified as a non-essential (conditionally essential) amino acid because it can be made from phenylalanine, provided the conversion enzyme is functioning.
The reaction is:
Phenylalanine + O₂ + BH₄ → Tyrosine + BH₂ + H₂O
- Enzyme: Phenylalanine hydroxylase (PAH) — a mixed-function monooxygenase
- Cofactor: Tetrahydrobiopterin (BH₄, also written as THB) — acts as an electron donor
- Location: Primarily in the liver (cytoplasm)
- Rate: This is a fast, high-capacity reaction under normal conditions — hence the MCQ phrase “quickly converted”
The hydroxylation adds a hydroxyl (-OH) group to the benzene ring of phenylalanine, converting it into tyrosine (which has a para-hydroxyl group on its ring — hence the “para” in para-aminobenzoic acid analogies). BH₄ is oxidised to BH₂ (dihydrobiopterin) in the process. BH₂ is then regenerated back to BH₄ by dihydropteridine reductase (DHPR) using NADH.
Think of BH₄ as a rechargeable battery — it gets “used up” in the hydroxylation but recharged by DHPR.
1B. Why Tyrosine Becomes “Conditionally Essential”
When PAH is deficient (as in PKU), the body can no longer make tyrosine from phenylalanine. Tyrosine therefore becomes diet-essential — it must now be supplied in food. This is why PKU dietary management includes not just phenylalanine restriction, but also tyrosine supplementation.
The other amino acids in the MCQ options (Arginine, Glycine, Leucine) have no direct metabolic connection to tyrosine biosynthesis:
- Arginine is part of the urea cycle and makes nitric oxide, ornithine, creatine
- Glycine is the simplest amino acid — it participates in heme synthesis, conjugation reactions, one-carbon metabolism
- Leucine is a branched-chain, purely ketogenic amino acid — metabolised entirely to acetyl-CoA and acetoacetate
🏥 Section 2 — The Downstream Metabolic Fate of Tyrosine
Once formed, tyrosine sits at a major metabolic crossroads. Depending on the tissue and physiological state, it can be directed into four major pathways:
2A. Catecholamine Synthesis (Adrenal Medulla & Brain)
Tyrosine → DOPA (via tyrosine hydroxylase, requires BH₄) → Dopamine → Norepinephrine → Epinephrine
This pathway is critical in the adrenal medulla and dopaminergic/noradrenergic neurons. Tyrosine hydroxylase is the rate-limiting enzyme of catecholamine synthesis. Inhibited by metyrosine (used to treat pheochromocytoma before surgery).
2B. Thyroid Hormone Synthesis (Thyroid Gland)
Tyrosine residues in thyroglobulin are iodinated to form:
- Monoiodotyrosine (MIT)
- Diiodotyrosine (DIT)
- Coupling → T₃ (MIT + DIT) and T₄ (DIT + DIT)
This is why iodine deficiency impairs thyroid hormone production and why pendred syndrome (defective iodine organification) affects this step.
2C. Melanin Synthesis (Melanocytes)
Tyrosine → DOPA (via tyrosinase, a copper-containing enzyme) → Dopaquinone → → → Melanin
Deficiency of tyrosinase → Oculocutaneous Albinism Type 1 (OCA1) — the classic white hair, pink eyes, pale skin presentation. Note: Albinism does NOT affect intelligence because melanin is not needed for brain development.
2D. Catabolism — Fumarate and Acetoacetate
Tyrosine is both glucogenic AND ketogenic — it can be broken down to fumarate (enters TCA, glucogenic) and acetoacetate (ketogenic). The catabolic pathway goes through several intermediates including homogentisic acid, which is relevant to alkaptonuria.
🧪 Section 3 — Disorders of the Phenylalanine-Tyrosine Pathway
Phenylketonuria (PKU) — The Classic Disorder
Enzyme deficient: Phenylalanine hydroxylase (PAH)
Gene: PAH gene on chromosome 12q23.2
Inheritance: Autosomal recessive
Incidence: ~1 in 10,000–15,000 live births (varies by ethnicity)
Biochemistry: Phenylalanine accumulates → shunted to transamination → phenylpyruvate (a phenylketone) excreted in urine (hence “phenylketonuria”) → also forms phenylacetate and phenyllactate.
Clinical features:
- Normal at birth (protected in utero by maternal enzymes)
- Progressive intellectual disability if untreated
- Microcephaly
- Seizures (often infantile spasms)
- Hypopigmentation (fair hair, blue eyes, pale skin — because phenylalanine competes with tyrosine for tyrosinase)
- Eczema
- Musty/mousy urine odour (phenylacetate)
- Normal or decreased tyrosine levels
Diagnosis:
- Newborn screening: Guthrie bacterial inhibition assay (historically) or tandem mass spectrometry (current gold standard)
- Plasma phenylalanine > 120 μmol/L (normal < 120 μmol/L)
- Urine ferric chloride test: green colour (phenylpyruvate)
Treatment:
- Low phenylalanine diet (restrict natural protein, use special amino acid formula)
- Tyrosine supplementation
- Sapropterin (BH₄ analogue) — for BH₄-responsive PKU (~25–50% of patients)
- Maternal PKU: strict diet control before and during pregnancy (maternal PKU → teratogenic effects on fetus even if fetus is heterozygous)
Malignant (Atypical) PKU — BH₄ Deficiency
Enzyme deficient: Dihydropteridine reductase (DHPR) or enzymes in BH₄ synthesis
Key distinction: Phenylalanine levels elevated (like classic PKU) BUT low-phenylalanine diet does NOT prevent neurological deterioration
Why: BH₄ is also required for tyrosine hydroxylase and tryptophan hydroxylase — deficiency depletes dopamine and serotonin
Treatment: BH₄ supplementation + neurotransmitter precursors (L-DOPA, 5-hydroxytryptophan)
Alkaptonuria
Enzyme deficient: Homogentisic acid oxidase
Pathway: Tyrosine catabolism
Biochemistry: Homogentisic acid accumulates → polymerises in tissues
Classic triad:
- Dark urine (on standing or alkalinisation — homogentisic acid oxidises to a dark pigment)
- Ochronosis (bluish-black pigmentation of cartilage, connective tissue, sclera)
- Arthritis (degenerative, particularly of spine and large joints — pigment deposits damage cartilage)
Inheritance: Autosomal recessive
Diagnosis: Urine turns dark on standing; urine reducing substance positive but glucose negative; confirmed by urine organic acids
Treatment: Nitisinone (NTBC) — inhibits 4-hydroxyphenylpyruvate dioxygenase (reduces homogentisic acid production); high dose Vitamin C
Tyrosinemia — Three Types
| Type | Deficient Enzyme | Key Feature |
|---|---|---|
| Type I (Hereditary) | Fumarylacetoacetate hydrolase | Severe liver failure, renal tubular dysfunction, hepatocellular carcinoma risk; “cabbage-like” odour; treated with nitisinone |
| Type II (Richner-Hanhart) | Tyrosine aminotransferase | Corneal erosions, palmar/plantar keratosis, intellectual disability |
| Type III | 4-hydroxyphenylpyruvate dioxygenase | Mild neurological problems; rare |
Albinism
OCA Type 1: Tyrosinase deficient (copper-enzyme) — complete lack of melanin
OCA Type 2: P-protein deficient — reduced melanin (most common type worldwide)
OCA Type 4: MATP transporter deficient
Ocular albinism (OA1): X-linked, affects only eyes
💊 Section 4 — Management Principles
PKU dietary management:
- Phenylalanine is essential — cannot be eliminated completely; must be restricted to safe levels
- Target plasma phenylalanine: 120–360 μmol/L (some guidelines 120–600 μmol/L for older patients)
- “Natural protein” severely restricted; medical protein formula (phenylalanine-free amino acid supplement) is the main protein source
- High-phenylalanine foods to avoid: meat, fish, eggs, dairy, nuts, regular bread, pasta (all high-protein foods); also aspartame (contains phenylalanine — why products say “Phenylketonurics: contains phenylalanine”)
- Safe foods: fruits, some vegetables, low-protein speciality foods
Sapropterin (Kuvan):
- Synthetic BH₄
- Works only in “BH₄-responsive” PAH mutations (missense mutations with residual enzyme activity)
- Allows some dietary liberalisation
- Does NOT replace dietary management completely in most patients
🎯 High-Yield Exam Facts
These are the facts that appear repeatedly across NEET PG, USMLE, AIIMS and FMGE papers.
- 🔴 Phenylalanine → Tyrosine via Phenylalanine Hydroxylase (PAH) — requires BH₄ as cofactor; occurs in liver; this is THE high-yield fact of this topic
- 🔴 PKU: autosomal recessive, PAH deficiency, musty urine odour — intellectual disability + fair pigmentation + eczema = classic exam vignette
- 🔴 Tyrosine hydroxylase is rate-limiting for catecholamine synthesis — BH₄ required here too
- 🔴 Tyrosinase (copper-containing) is rate-limiting for melanin synthesis — deficiency = OCA Type 1
- 🔴 Alkaptonuria: homogentisic acid oxidase deficiency — dark urine + ochronosis + arthritis triad; first disease shown to follow Mendelian inheritance (Garrod, 1902)
- 🟠 Malignant PKU: normal diet restriction doesn’t help — BH₄ deficiency affects dopamine AND serotonin synthesis; needs neurotransmitter replacement
- 🟠 Tyrosinemia Type I: treated with nitisinone — inhibits upstream enzyme to prevent toxic metabolite accumulation; liver failure + renal tubular dysfunction
- 🟠 Maternal PKU — mother with PKU must maintain strict dietary control during pregnancy; her high phenylalanine crosses placenta and damages fetal brain (microcephaly, CHD, IUGR) even if fetus is heterozygous
- 🟡 Aspartame warning — aspartame (artificial sweetener) is metabolised to phenylalanine; dangerous for PKU patients; products must carry warning
- 🟡 Tyrosine is “conditionally essential” — non-essential only if phenylalanine and PAH are available; becomes essential in PKU
- 🟡 BH₄ regeneration by DHPR uses NADH — defect in DHPR causes malignant PKU; BH₄ supplementation is treatment
- 🟡 Guthrie test — bacterial inhibition assay using Bacillus subtilis; historically used for PKU newborn screening; now largely replaced by tandem mass spectrometry
🧠 Mnemonics & Memory Tricks
Mnemonic: “Please Help All Babies Test Healthy”
Stands for: PAH (Phenylalanine Hydroxylase) requires BH₄ (Tetrahydrobiopterin) → converts To Healthy tyrosine
Use it for: Remembering the enzyme and cofactor for the phenylalanine → tyrosine step
Mnemonic: “DAMN the tyrosine pathway!”
Stands for: Dopamine, Adrenaline (epinephrine), Melanin, thyroid hormones (N for iodine-containing/Not-mentioned = thyroid T3/T4)
Use it for: Remembering the four major products of tyrosine metabolism
Mnemonic: “OAK for Alkaptonuria”
Stands for: Ochronosis, Arthritis, darK urine
Use it for: The classic triad of alkaptonuria
Mnemonic: “PKU patients smell Mousy, look Fair, and think Slowly“
Use it for: Musty/mousy odour + hypopigmentation + intellectual disability = PKU clinical picture
⚠️ Common Mistakes Students Make
❌ Mistake: “Tyrosine is an essential amino acid”
✅ Reality: Tyrosine is non-essential (conditionally essential) — the body makes it from phenylalanine
📝 Exam trap: Questions ask “which of the following is non-essential?” — tyrosine is non-essential; phenylalanine is essential
❌ Mistake: “All PKU is treated with low-phenylalanine diet”
✅ Reality: Malignant/atypical PKU (BH₄ deficiency) does NOT respond to dietary phenylalanine restriction; needs BH₄ + neurotransmitter precursors
📝 Exam trap: Vignette says “PKU patient on low-Phe diet but still has neurological symptoms” → think malignant PKU/DHPR deficiency
❌ Mistake: “Albinism causes intellectual disability”
✅ Reality: Albinism (tyrosinase deficiency) causes absence of melanin pigment — it does NOT affect intelligence; no neurological involvement
📝 Exam trap: Don’t confuse albinism (OCA) with PKU; PKU has hypopigmentation but ALSO intellectual disability
❌ Mistake: “Alkaptonuria urine is always dark”
✅ Reality: Urine is normal coloured when fresh; it darkens on standing (exposure to air/alkalinisation causes oxidation of homogentisic acid)
📝 Exam trap: “Baby’s nappy/diaper turns black on standing” = alkaptonuria; not dark urine straight away
❌ Mistake: “BH₄ is only needed for phenylalanine hydroxylase”
✅ Reality: BH₄ is a cofactor for PAH, tyrosine hydroxylase, AND tryptophan hydroxylase — all three aromatic amino acid hydroxylases
📝 Exam trap: BH₄ deficiency causes problems beyond PKU — also depletes dopamine and serotonin; this is why malignant PKU is more severe
🔗 How This Topic Connects to Others
Understanding phenylalanine-tyrosine metabolism gives you automatic access to these tightly linked topics:
- Urea Cycle & Amino Acid Catabolism — Phenylalanine is both glucogenic and ketogenic; its complete catabolism connects to the TCA cycle
- Neurotransmitter Biochemistry — Dopamine, norepinephrine, and epinephrine are all derived from tyrosine; pharmacology of L-DOPA in Parkinson’s disease builds directly on this
- Thyroid Hormone Synthesis & Disorders — Tyrosine iodination is the foundation of T3/T4 biochemistry; connects to iodine deficiency goitre, Hashimoto’s, Graves’ disease
- Melanin & Skin Biology — Links to dermatology (vitiligo, albinism, Waardenburg syndrome) and oncology (melanoma, which overexpresses tyrosinase-related proteins as tumour antigens)
- Neonatal Screening Programs — PKU was the first disease included in universal newborn screening; connects to public health and preventive medicine; links to discussion of tandem mass spectrometry screening panels
❓ The MCQ That Started This — Fully Explained
Question: Which of the following amino acids is quickly converted to tyrosine?
- A. Arginine
- B. Glycine
- C. Phenylalanine
- D. Leucine
✅ Correct Answer: C. Phenylalanine
Why correct: Phenylalanine is converted to tyrosine by phenylalanine hydroxylase (PAH), a liver enzyme that uses BH₄ as a cofactor. This is a rapid, high-capacity hydroxylation reaction and is the sole biosynthetic route to tyrosine in the human body. The word “quickly” in the stem specifically signals this direct enzymatic conversion.
Why A is wrong: Arginine is part of the urea cycle and is a precursor for nitric oxide, ornithine, creatine, and polyamines — it has no metabolic pathway connecting it to tyrosine biosynthesis.
Why B is wrong: Glycine is the simplest amino acid (no chiral centre) and participates in heme synthesis (with succinyl-CoA), purine synthesis, and phase II conjugation reactions — it is not converted to tyrosine.
Why D is wrong: Leucine is a branched-chain amino acid and the only purely ketogenic amino acid among the essential amino acids — it is metabolised exclusively to acetyl-CoA and acetoacetate, with no connection to tyrosine.
📝 Test Your Understanding — 5 Practice MCQs
Q1. Which cofactor is required by phenylalanine hydroxylase for the conversion of phenylalanine to tyrosine?
- A. Pyridoxal phosphate (PLP)
- B. Tetrahydrobiopterin (BH₄)
- C. Tetrahydrofolate (THF)
- D. Flavin adenine dinucleotide (FAD)
✅ **B. Tetrahydrobiopterin (BH₄)** — BH₄ is the essential electron-donating cofactor for phenylalanine hydroxylase; it is oxidised to BH₂ in the reaction and regenerated by dihydropteridine reductase (DHPR) using NADH.
Q2. A deficiency of which enzyme is responsible for the accumulation of homogentisic acid?
- A. Phenylalanine hydroxylase
- B. Tyrosinase
- C. Homogentisic acid oxidase
- D. Fumarylacetoacetate hydrolase
✅ **C. Homogentisic acid oxidase** — Alkaptonuria results from deficiency of this enzyme in the tyrosine catabolism pathway. Homogentisic acid accumulates, causing the classic triad of dark urine (on standing), ochronosis, and arthritis.
Q3. A patient with phenylketonuria who is maintained on a strict low-phenylalanine diet still develops progressive neurological deterioration with low CSF levels of dopamine and serotonin metabolites. The MOST likely additional enzyme deficiency is:
- A. Tyrosine aminotransferase
- B. Dihydropteridine reductase (DHPR)
- C. Fumarylacetoacetate hydrolase
- D. Homogentisic acid oxidase
✅ **B. Dihydropteridine reductase (DHPR)** — This is malignant/atypical PKU. DHPR deficiency means BH₄ cannot be regenerated, depleting tetrahydrobiopterin needed for tyrosine hydroxylase and tryptophan hydroxylase as well — hence dopamine AND serotonin deficiency despite dietary phenylalanine control.
Q4. A 28-year-old woman with previously well-controlled PKU becomes pregnant. Her obstetric team advises strict dietary phenylalanine restriction throughout pregnancy. The PRIMARY reason for this advice is:
- A. The fetus inherits PAH deficiency and cannot clear phenylalanine
- B. High maternal phenylalanine crosses the placenta and is teratogenic to the fetus regardless of fetal genotype
- C. Phenylalanine competes with folic acid absorption, causing neural tube defects
- D. Phenylalanine excess causes maternal hypoglycaemia during pregnancy
✅ **B. High maternal phenylalanine crosses the placenta and is teratogenic to the fetus regardless of fetal genotype** — Maternal PKU syndrome causes microcephaly, intellectual disability, congenital heart defects, and IUGR in the offspring even if the fetus is only a heterozygous carrier. Strict dietary control must begin before conception.
Q5. A diet drink carries the label: “Phenylketonurics: Contains Phenylalanine.” The sweetener responsible for this warning is:
- A. Saccharin
- B. Sucralose
- C. Aspartame
- D. Stevia
✅ **C. Aspartame** — Aspartame (NutraSweet, Equal) is a dipeptide of aspartic acid and phenylalanine methyl ester. When metabolised, it releases phenylalanine, which is dangerous for PKU patients. All products containing aspartame are legally required to carry the phenylketonurics warning in most countries.
📚 References & Further Reading
- Harper’s Illustrated Biochemistry — 32nd Edition; Chapter 29: Catabolism of Proteins & Amino Acid Nitrogen; Chapter 30: Conversion of Amino Acids to Specialised Products
- Lippincott’s Illustrated Reviews: Biochemistry — 7th Edition; Chapter 20: Amino Acid Degradation and Synthesis; Chapter 21: Conversion of Amino Acids to Specialised Products
- Robbins & Cotran Pathologic Basis of Disease — 10th Edition; Chapter 5: Genetic Disorders — Inborn Errors of Metabolism (PKU, Alkaptonuria, Albinism)
- Nelson Textbook of Pediatrics — 21st Edition; Chapter 103: Defects in Metabolism of Amino Acids (Phenylalanine, Tyrosine disorders)
- Biochemistry — Stryer, Berg & Tymoczko; 9th Edition; Chapter 23: Protein Turnover and Amino Acid Catabolism
🚀 Ready to Master Biochemistry?
You’ve just covered one of the most high-yield topics in Biochemistry — the phenylalanine-tyrosine metabolic axis. The pathway, the disorders, the clinical presentations, and the exam traps are now all mapped in your mind.
medicalmcq.in has hundreds of free Biochemistry MCQs — each with detailed explanations just like this article.
When you’re ready to simulate real exam pressure, our Mock Test Series gives you timed, subject-wise and full-length tests with performance analytics — so you know exactly where to focus next.