What You Will Learn in This Article
- Recall all 10 steps of glycolysis in order, with the correct enzyme for each step
- Identify the 3 irreversible steps and why they matter for regulation
- State the exact ATP investment, ATP yield, and net gain from one glucose molecule
- Name all 3 rate-limiting enzymes and the factors that activate or inhibit each one
- Connect glycolysis to gluconeogenesis, the TCA cycle, and the pentose phosphate pathway
- Recognise clinical diseases caused by glycolytic enzyme deficiencies
- Answer both recall-type and clinical-scenario MCQs on this topic with confidence
📖 Introduction: Why This Topic Matters in Exams
Imagine a patient presenting with chronic haemolytic anaemia. No immune cause is found. The Coombs test is negative. The haematologist orders red blood cell enzyme assays — and the culprit turns out to be a deficiency of pyruvate kinase, the final enzyme of glycolysis. Red blood cells have no mitochondria — glycolysis is their only source of ATP. When it fails, they cannot maintain their membrane integrity, and they lyse. This is not a textbook curiosity. It is the kind of clinical reasoning that NEET PG and USMLE Step 1 test directly.
Glycolysis appears in virtually every major entrance exam, and it appears in multiple forms — pure recall questions (“which enzyme is rate-limiting?”), mechanism questions (“why does arsenate poisoning cause ATP depletion?”), and clinical-scenario questions (“a child with haemolytic anaemia has elevated 2,3-BPG — which enzyme is deficient?”). It is one of the highest-yield topics in biochemistry, and the students who score on it are the ones who know every step, every enzyme, and every regulation point — not just the broad outline.
This article takes you through all 10 steps of glycolysis with every enzyme named, every product accounted for, the full regulatory framework, the clinical connections, and a set of practice MCQs to test whether you actually retained it. Read it once carefully, then do the questions at the end. That combination will lock this topic into long-term memory.
🔬 Section 1 — The 10 Steps of Glycolysis, Explained Step by Step
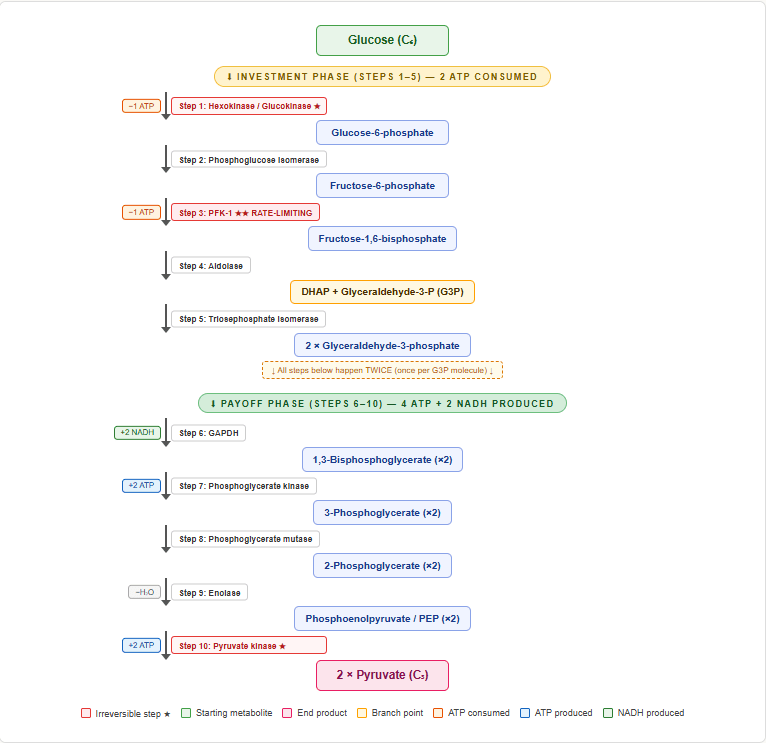
Overview: The Big Picture Before the Details
Glycolysis is the universal pathway for the breakdown of glucose to pyruvate. It occurs in the cytoplasm of every living cell. It does not require oxygen — it is anaerobic — which makes it essential in tissues with limited oxygen supply such as red blood cells, the renal medulla, and exercising skeletal muscle.
The pathway has two distinct phases:
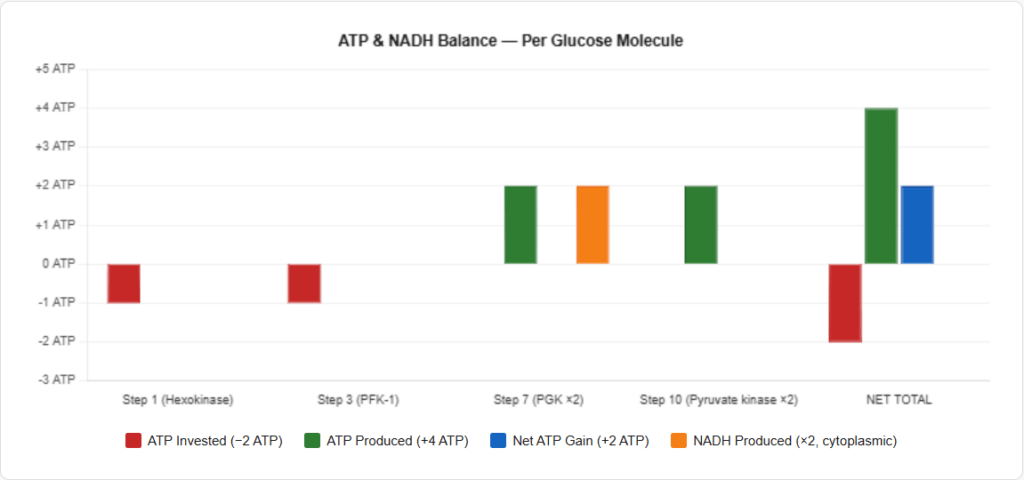
- Investment phase (Steps 1–5): The cell spends 2 ATP to phosphorylate and split glucose into two 3-carbon units
- Payoff phase (Steps 6–10): Each 3-carbon unit generates 2 ATP and 1 NADH, giving a total of 4 ATP and 2 NADH
Net yield from one glucose molecule:
- Net ATP = 4 produced − 2 invested = 2 ATP
- NADH = 2 NADH (in the cytoplasm)
- Pyruvate = 2 molecules
The 10 Steps — Every Enzyme, Every Product
STEP 1 — Glucose → Glucose-6-phosphate
- Enzyme: Hexokinase (in most tissues) / Glucokinase (in liver and pancreatic β-cells)
- Cofactor: Mg²⁺, requires ATP (1 ATP consumed)
- Irreversible — this is the first committed step
- Key distinction: Hexokinase has a low Km (high affinity, works even at low glucose). Glucokinase has a high Km (low affinity, only active when glucose is high — acts as a glucose sensor in the liver)
- Glucose-6-phosphate cannot cross the cell membrane — this step traps glucose inside the cell
STEP 2 — Glucose-6-phosphate → Fructose-6-phosphate
- Enzyme: Phosphoglucose isomerase (Phosphohexose isomerase)
- Reversible — this is an isomerisation reaction
- Converts an aldose (glucose-6-P) to a ketose (fructose-6-P)
STEP 3 — Fructose-6-phosphate → Fructose-1,6-bisphosphate
- Enzyme: Phosphofructokinase-1 (PFK-1) — the rate-limiting enzyme of glycolysis
- Cofactor: Mg²⁺, requires ATP (1 ATP consumed — total investment now 2 ATP)
- Irreversible — the most important regulatory step in the entire pathway
- PFK-1 is the master on/off switch of glycolysis
STEP 4 — Fructose-1,6-bisphosphate → DHAP + Glyceraldehyde-3-phosphate
- Enzyme: Aldolase
- Reversible — cleaves the 6-carbon sugar into two 3-carbon molecules:
- Dihydroxyacetone phosphate (DHAP)
- Glyceraldehyde-3-phosphate (G3P)
- Only G3P directly enters the next step; DHAP is converted to G3P by the next enzyme
STEP 5 — DHAP → Glyceraldehyde-3-phosphate
- Enzyme: Triosephosphate isomerase (TPI)
- Reversible — converts DHAP to G3P
- After this step, both 3-carbon units are G3P, so all subsequent steps happen twice per glucose molecule
- TPI deficiency causes haemolytic anaemia + neuromuscular disease
End of the Investment Phase — The cell has spent 2 ATP and now has 2 molecules of G3P
STEP 6 — Glyceraldehyde-3-phosphate → 1,3-Bisphosphoglycerate
- Enzyme: Glyceraldehyde-3-phosphate dehydrogenase (GAPDH)
- Reversible
- This step oxidises G3P and simultaneously phosphorylates it using inorganic phosphate (not ATP)
- 1 NADH produced per G3P (× 2 = 2 NADH total from one glucose)
- This is the step blocked by arsenate (arsenate substitutes for phosphate, forming an unstable product that cannot generate ATP downstream — net ATP yield drops to zero)
- Iodoacetate also inhibits GAPDH (used in lab experiments to block glycolysis)
STEP 7 — 1,3-Bisphosphoglycerate → 3-Phosphoglycerate
- Enzyme: Phosphoglycerate kinase (PGK)
- Reversible
- 1 ATP generated per molecule (× 2 = 2 ATP — the investment of 2 ATP is now recovered)
- This is an example of substrate-level phosphorylation
- 2,3-Bisphosphoglycerate (2,3-BPG) is produced from 1,3-BPG by the Rapoport-Luebering shunt in red blood cells — this is the allosteric regulator of haemoglobin oxygen affinity
STEP 8 — 3-Phosphoglycerate → 2-Phosphoglycerate
- Enzyme: Phosphoglycerate mutase
- Reversible — moves the phosphate group from carbon 3 to carbon 2
- Requires 2,3-BPG as a cofactor (a fact occasionally tested)
STEP 9 — 2-Phosphoglycerate → Phosphoenolpyruvate (PEP)
- Enzyme: Enolase
- Reversible
- Removes a water molecule (dehydration reaction)
- Inhibited by fluoride — this is why fluoride is added to blood collection tubes for glucose measurement (it inhibits enolase and stops red cells from consuming glucose in the tube)
STEP 10 — Phosphoenolpyruvate → Pyruvate
- Enzyme: Pyruvate kinase (PK)
- Irreversible — the third and final irreversible step
- 1 ATP generated per molecule (× 2 = 2 ATP — making gross total 4 ATP, net 2 ATP)
- Substrate-level phosphorylation
- Pyruvate kinase deficiency is the most common cause of hereditary non-spherocytic haemolytic anaemia
Summary Table: All 10 Steps at a Glance
| Step | Substrate | Product | Enzyme | ATP Change | Reversible? |
|---|---|---|---|---|---|
| 1 | Glucose | Glucose-6-P | Hexokinase / Glucokinase | −1 ATP | No |
| 2 | Glucose-6-P | Fructose-6-P | Phosphoglucose isomerase | — | Yes |
| 3 | Fructose-6-P | Fructose-1,6-bisP | PFK-1 (rate-limiting) | −1 ATP | No |
| 4 | Fructose-1,6-bisP | DHAP + G3P | Aldolase | — | Yes |
| 5 | DHAP | G3P | Triosephosphate isomerase | — | Yes |
| 6 | G3P | 1,3-Bisphosphoglycerate | GAPDH | +2 NADH | Yes |
| 7 | 1,3-BPG | 3-Phosphoglycerate | Phosphoglycerate kinase | +2 ATP | Yes |
| 8 | 3-Phosphoglycerate | 2-Phosphoglycerate | Phosphoglycerate mutase | — | Yes |
| 9 | 2-Phosphoglycerate | PEP | Enolase | — | Yes |
| 10 | PEP | Pyruvate | Pyruvate kinase | +2 ATP | No |
(Steps 6–10 yield are per glucose, i.e. ×2 because two G3P molecules are processed)
⚙️ Section 2 — Regulation of Glycolysis: The 3 Key Control Points
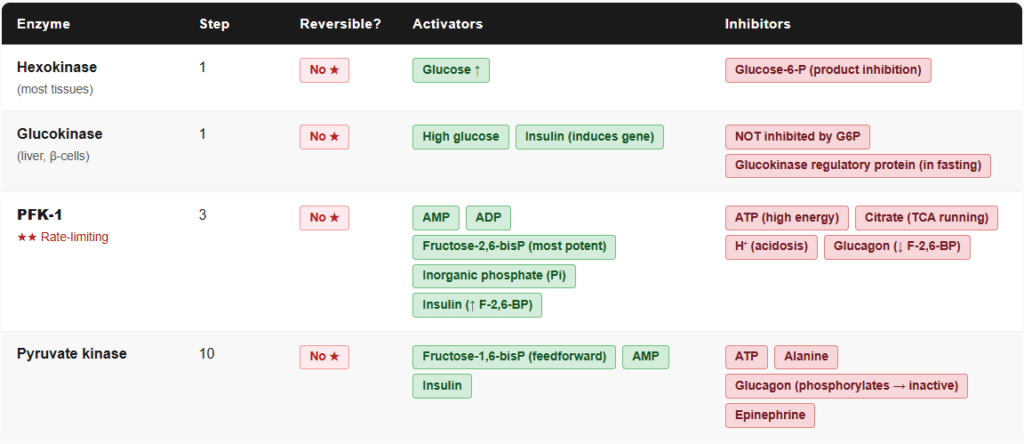
The three irreversible steps of glycolysis are also the three regulated steps. Regulation happens at Steps 1, 3, and 10.
Control Point 1: Hexokinase / Glucokinase (Step 1)
Hexokinase (most tissues):
- Inhibited by its own product: Glucose-6-phosphate (product inhibition)
- This prevents the cell from trapping more glucose than it can use
Glucokinase (liver and pancreatic β-cells):
- NOT inhibited by glucose-6-phosphate
- Has sigmoidal kinetics — responds proportionally to rising glucose concentration
- Acts as a glucose sensor: it only becomes significantly active after a meal when portal glucose rises
- Induced by insulin — this is why liver glycolysis increases after eating
Control Point 2: PFK-1 (Step 3) — The Master Switch
PFK-1 is the most important regulatory enzyme in glycolysis. Think of it as the throttle.
| Activators of PFK-1 | Inhibitors of PFK-1 |
|---|---|
| AMP (low energy signal) | ATP (high energy — no need to make more) |
| ADP | Citrate (TCA cycle running well) |
| Fructose-2,6-bisphosphate (F-2,6-BP) — most potent | H⁺ (acidosis — prevents ATP depletion in ischaemia) |
| Pi (inorganic phosphate) | Glucagon (via lowering F-2,6-BP) |
| Insulin (via raising F-2,6-BP) |
Fructose-2,6-bisphosphate (F-2,6-BP) is the most potent allosteric activator of PFK-1 and is controlled by a bifunctional enzyme called PFK-2/FBPase-2:
- Insulin → activates PFK-2 → raises F-2,6-BP → PFK-1 activated → glycolysis increases
- Glucagon → activates FBPase-2 → lowers F-2,6-BP → PFK-1 inhibited → glycolysis slows
This is the hormonal switch that tells the liver to either break down glucose (fed state) or spare it (fasting state).
Control Point 3: Pyruvate Kinase (Step 10)
| Activators | Inhibitors |
|---|---|
| Fructose-1,6-bisphosphate (feedforward activation) | ATP |
| AMP | Alanine (amino acid signal — precursor available) |
| Glucagon (via phosphorylation — inactivates PK) | |
| Epinephrine |
Feedforward activation by fructose-1,6-bisphosphate is elegant: when PFK-1 is active and producing F-1,6-BP, that same molecule goes ahead and pre-activates pyruvate kinase, ensuring the pathway downstream is ready to handle the flux.
🏥 Section 3 — Clinical Connections: When Glycolysis Goes Wrong
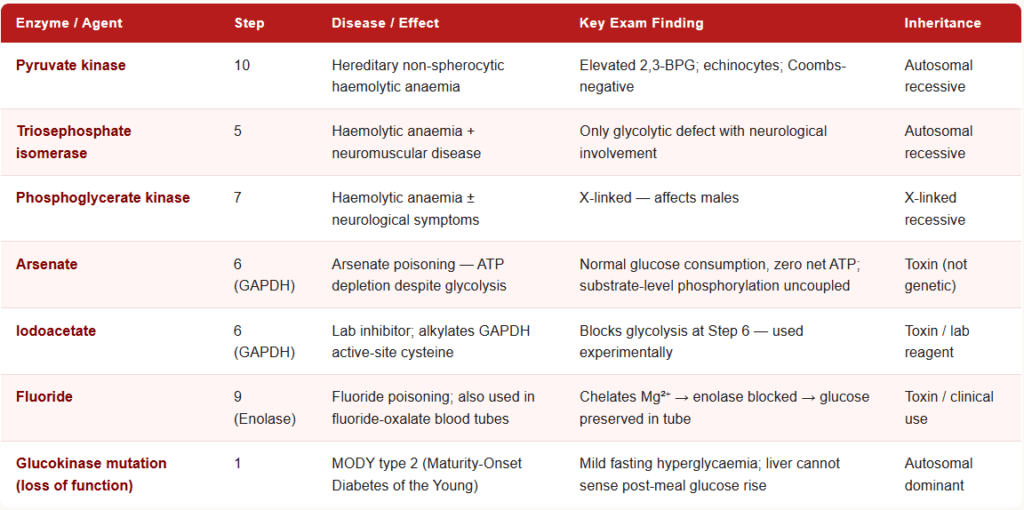
Pyruvate Kinase Deficiency
The most common hereditary non-spherocytic haemolytic anaemia (after G6PD deficiency, which is in the pentose phosphate pathway).
- Inheritance: Autosomal recessive
- Mechanism: PK deficiency → cannot complete Step 10 → ATP depletion in RBCs → RBC membrane fails → haemolysis
- Key finding: Elevated 2,3-BPG (because 1,3-BPG, which cannot be converted to pyruvate, backs up into the Rapoport-Luebering shunt)
- Elevated 2,3-BPG causes a right shift of the oxygen-dissociation curve (reduced haemoglobin-oxygen affinity — actually partially compensatory)
- Peripheral smear: echinocytes (spiculated RBCs)
- Exam tip: “Haemolytic anaemia + elevated 2,3-BPG + Coombs-negative” = Pyruvate kinase deficiency
Triosephosphate Isomerase (TPI) Deficiency
- Rare but high-yield because of its unusual presentation
- Causes haemolytic anaemia PLUS progressive neuromuscular disease (the only glycolytic enzyme deficiency with neurological involvement)
- Autosomal recessive
Arsenate Poisoning
- Arsenate (AsO₄³⁻) is a structural analogue of phosphate
- At Step 6, GAPDH incorporates arsenate instead of phosphate → forms 1-arseno-3-phosphoglycerate, which spontaneously hydrolyses
- The high-energy bond is lost before ATP can be generated at Step 7
- Result: Glycolysis continues but generates no net ATP — the cell burns glucose for nothing
- Arsenate uncouples substrate-level phosphorylation
Fluoride Poisoning (Iodoacetate — Lab Context)
- Fluoride inhibits enolase (Step 9) by chelating Mg²⁺
- Used clinically: fluoride-oxalate tubes for blood glucose measurement preserve the sample
- Iodoacetate inhibits GAPDH (Step 6) by alkylating the active-site cysteine
Glucose-6-phosphate Dehydrogenase (G6PD) Deficiency
- Strictly speaking this is a pentose phosphate pathway enzyme, not a glycolytic enzyme — but it is often tested alongside glycolysis because the two pathways branch at glucose-6-phosphate
- G6PD deficiency → no NADPH → cannot regenerate glutathione → oxidative haemolysis with Heinz bodies
🔄 Section 4 — How Glycolysis Connects to the Rest of Metabolism
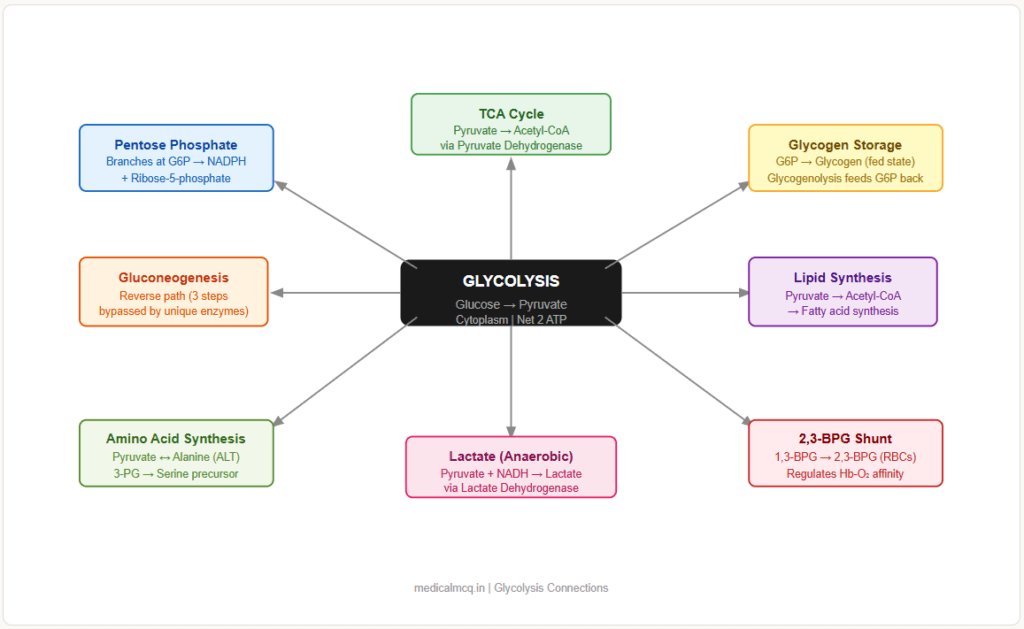
Understanding glycolysis in isolation is not enough — exams increasingly test integrated metabolism.
→ Gluconeogenesis — The reverse of glycolysis, but the three irreversible steps are bypassed by four unique enzymes (pyruvate carboxylase, PEPCK, fructose-1,6-bisphosphatase, glucose-6-phosphatase). Knowing which steps are bypassed tells you exactly which enzymes are unique to gluconeogenesis.
→ TCA Cycle (Krebs Cycle) — Pyruvate (the end product of glycolysis) is converted to Acetyl-CoA by pyruvate dehydrogenase complex before entering the TCA cycle. Glycolysis feeds the TCA cycle in aerobic conditions.
→ Pentose Phosphate Pathway (PPP) — Branches at glucose-6-phosphate (after Step 1). The PPP generates NADPH and ribose-5-phosphate. When PPP flux is high, G6P is diverted away from glycolysis.
→ Glycogen Synthesis / Glycogenolysis — Also branches at glucose-6-phosphate. In the fed state, G6P can go to glycogen storage. In fasting, glycogenolysis releases G6P back into glycolysis.
→ Lipid Synthesis — Pyruvate → Acetyl-CoA → fatty acid synthesis. Excess glucose carbons (via glycolysis) are the carbon source for de novo lipogenesis. This is why hyperglycaemia drives hypertriglyceridaemia.
→ Amino Acid Metabolism — Pyruvate is in equilibrium with alanine (via alanine aminotransferase). 3-phosphoglycerate is a precursor for serine. The glycolytic intermediate DHAP connects to glycerophospholipid synthesis.
→ Lactate Metabolism — When oxygen is insufficient (anaerobic conditions), pyruvate is reduced to lactate by lactate dehydrogenase (LDH), regenerating NAD⁺ so glycolysis can continue. This is the basis of lactic acidosis in shock.
🎯 High-Yield Exam Facts
These are the exact facts that appear repeatedly across NEET PG, USMLE, AIIMS and FMGE papers.
Read these. Memorise these. These are your marks.
🔴 PFK-1 is the rate-limiting enzyme of glycolysis
Not hexokinase, not pyruvate kinase — PFK-1 is the answer every time a question asks for the “rate-limiting” or “regulatory” enzyme of glycolysis.
🔴 Three irreversible steps: Steps 1, 3, and 10 (Hexokinase, PFK-1, Pyruvate kinase)
These are also the three steps that are bypassed in gluconeogenesis by different enzymes.
🔴 Net ATP yield from one glucose = 2 ATP (anaerobic); 30–32 ATP (aerobic)
In glycolysis alone: 4 ATP produced − 2 ATP invested = 2 net ATP. The 2 NADH from Step 6 yield additional ATP only if oxygen is available for the electron transport chain.
🔴 Glucokinase has high Km and is NOT inhibited by its product (G6P)
This is the opposite of hexokinase. Glucokinase acts as a liver glucose sensor — only activated by high post-meal glucose. Induced by insulin.
🟠 Fructose-2,6-bisphosphate (F-2,6-BP) is the most potent activator of PFK-1
It is not a glycolytic intermediate itself — it is a regulatory molecule made by PFK-2. Insulin raises it; glucagon lowers it.
🟠 Fluoride inhibits enolase (Step 9)
Mechanism: fluoride chelates the Mg²⁺ required by enolase. This is why fluoride-oxalate tubes preserve blood glucose — they stop RBCs from consuming glucose in vitro.
🟠 Arsenate poisoning uncouples substrate-level phosphorylation at Step 6 (GAPDH)
Arsenate substitutes for phosphate, forming an unstable product. ATP cannot be generated at Step 7. The result: normal glycolysis but zero net ATP.
🟠 Pyruvate kinase deficiency → elevated 2,3-BPG → right shift of O₂ dissociation curve
The backup of 1,3-BPG into the Rapoport-Luebering shunt produces excess 2,3-BPG, which reduces Hb-O₂ affinity.
🟡 Aldolase B deficiency causes hereditary fructose intolerance, NOT a glycolytic defect
Aldolase B works in fructose metabolism. Aldolase A is the glycolytic enzyme. Do not confuse them.
🟡 Iodoacetate inhibits GAPDH; arsenate also acts at GAPDH — but by different mechanisms
Iodoacetate: alkylates the active-site cysteine (irreversible). Arsenate: mimics phosphate (competes for the active site). Both block Step 6 — but the question will phrase them differently.
🟡 TPI deficiency is the only glycolytic enzyme deficiency with neurological involvement
All other glycolytic enzyme deficiencies cause haemolysis only (RBCs are most vulnerable because they have no mitochondria). TPI deficiency adds progressive neuromuscular disease.
🟡 Pyruvate kinase activity is stimulated by its own substrate’s precursor: F-1,6-bisphosphate
This is feedforward (not feedback) activation — the incoming signal activates the downstream enzyme before the substrate actually arrives.
🧠 Mnemonics & Memory Tricks
“Horrible People Fight And Take Good People Home Every Paycheck”
→ Helps you remember: The 10 enzymes of glycolysis in order
H = Hexokinase (Step 1)
P = Phosphoglucose isomerase (Step 2)
F = PFK-1 (Step 3)
A = Aldolase (Step 4)
T = Triosephosphate isomerase (Step 5)
G = GAPDH (Step 6)
P = Phosphoglycerate kinase (Step 7)
H = Phosphoglycerate mutase (Step 8 — H for “Has moved”)
E = Enolase (Step 9)
P = Pyruvate kinase (Step 10)
💡 Pro tip: When a question asks “which enzyme acts at Step 7?” count along this sentence — the 7th word is “Good” = Phosphoglycerate kinase.
“Always Inhibit PFK-1 with ATP and Citrate”
→ Helps you remember: The two major inhibitors of PFK-1
ATP = High energy state → no need for more glycolysis
Citrate = TCA cycle is running well → slow down the fuel supply
💡 Pro tip: When a question gives a scenario of a “well-fed cell with plenty of oxygen”, both ATP and citrate will be high → PFK-1 is inhibited → glycolysis is turned off. This is the correct reasoning chain.
“GlucoKinase is a Km-High, insulin-Kindled glucose-sensor in the Liver”
→ Helps you remember the 4 key features of glucokinase vs hexokinase
Km-High = low affinity (opposite of hexokinase)
insulin-Kindled = induced by insulin
glucose-sensor = only active at high glucose (post-meal)
Liver (and β-cells) = where it is expressed
💡 Pro tip: Any question with “liver glucose sensing”, “post-meal glucose uptake”, or “insulin induction of glycolysis” is pointing at glucokinase.
⚠️ Common Mistakes Students Make on This Topic
❌ Mistake: “Hexokinase is the rate-limiting enzyme of glycolysis”
✅ Reality: PFK-1 is the rate-limiting enzyme. Hexokinase catalyses the first committed step, but the rate-limiting (slowest, most regulated) step is Step 3.
📝 How this gets tested: Options will list both Hexokinase and PFK-1. Students who confuse “first committed step” with “rate-limiting step” choose hexokinase and lose the mark.
❌ Mistake: “Glycolysis occurs in mitochondria”
✅ Reality: Glycolysis occurs entirely in the cytoplasm (cytosol). Only pyruvate dehydrogenase and the TCA cycle are mitochondrial.
📝 How this gets tested: Questions on subcellular location. The trap: because aerobic glucose oxidation ultimately uses mitochondria, students incorrectly place glycolysis there too.
❌ Mistake: “Fructose-2,6-bisphosphate is a glycolytic intermediate”
✅ Reality: F-2,6-BP is a regulatory molecule, not a pathway intermediate. It is not produced during glycolysis — it is made separately by PFK-2 solely to regulate PFK-1.
📝 How this gets tested: Questions asking “which of the following is a product of glycolysis?” — F-2,6-BP is a tempting trap if you confuse it with fructose-1,6-bisphosphate (which IS a true intermediate).
❌ Mistake: “Arsenate poisoning stops glycolysis”
✅ Reality: Arsenate does NOT stop glycolysis — it uncouples ATP generation from glycolysis. The pathway still runs and glucose is still consumed, but no net ATP is made.
📝 How this gets tested: “A patient exposed to arsenate shows increased glucose consumption but cellular energy failure” — the answer is not “glycolysis is blocked” but “substrate-level phosphorylation is uncoupled.”
❌ Mistake: “Pyruvate kinase deficiency causes spherocytosis”
✅ Reality: PK deficiency causes non-spherocytic haemolytic anaemia (echinocytes/spiculated cells). Spherocytosis is caused by spectrin/ankyrin defects. Both are Coombs-negative haemolytic anaemias, but morphology differs.
📝 How this gets tested: “Coombs-negative haemolytic anaemia” questions will distinguish by RBC morphology and 2,3-BPG levels.
📝 5 Practice MCQs — Test Yourself Now
Q1: Which enzyme catalyses the rate-limiting step of glycolysis?
- A. Hexokinase
- B. Pyruvate kinase
- C. Phosphofructokinase-1
- D. Aldolase
✅ Answer: C. Phosphofructokinase-1
Why correct: PFK-1 catalyses Step 3 (Fructose-6-phosphate → Fructose-1,6-bisphosphate) and is the rate-limiting, most heavily regulated enzyme of glycolysis. It is the committed, irreversible step at which the cell decides to proceed with glycolysis.
Why A is wrong: Hexokinase catalyses the first step and is the first committed step, but it is NOT the rate-limiting enzyme — PFK-1 is slower and more regulated.
Why B is wrong: Pyruvate kinase catalyses the final irreversible step (Step 10) and is regulated, but it is not the rate-limiting enzyme.
Why D is wrong: Aldolase catalyses a reversible cleavage reaction (Step 4) and has no rate-limiting role.
Exam tip: “Rate-limiting,” “regulatory,” and “committed step” are often used interchangeably in glycolysis questions. All three point to PFK-1 for glycolysis.
Q2: A patient’s blood sample is collected in a fluoride-oxalate tube. What is the biochemical basis for fluoride preventing falsely low glucose readings?
- A. Fluoride inhibits hexokinase, preventing glucose phosphorylation
- B. Fluoride inhibits enolase by chelating Mg²⁺, halting glycolysis at Step 9
- C. Fluoride inhibits GAPDH by alkylating its active-site cysteine
- D. Fluoride activates glucose-6-phosphatase, releasing free glucose
✅ Answer: B. Fluoride inhibits enolase by chelating Mg²⁺, halting glycolysis at Step 9
Why correct: Fluoride forms a complex with Mg²⁺ and inorganic phosphate that binds to and inhibits enolase (Step 9: 2-phosphoglycerate → PEP). This blocks glycolysis in the RBCs of the collected blood sample, preventing them from consuming the glucose being measured.
Why A is wrong: Hexokinase inhibition would also prevent glucose consumption, but this is NOT the mechanism of fluoride — it is product inhibition by G6P that regulates hexokinase.
Why C is wrong: Alkylation of GAPDH’s active-site cysteine is the mechanism of iodoacetate, not fluoride.
Why D is wrong: Glucose-6-phosphatase activation would release glucose from G6P, which is the opposite scenario — and fluoride has no such action.
Exam tip: “Fluoride tube” questions always test enolase. The secondary fact — that fluoride chelates Mg²⁺ — is the mechanism detail that distinguishes it from iodoacetate.
Q3: Under aerobic conditions in a well-fed hepatocyte, which combination of signals would maximally inhibit glycolysis at the PFK-1 step?
- A. High AMP + high fructose-2,6-bisphosphate
- B. High ATP + high citrate + low fructose-2,6-bisphosphate
- C. High insulin + high AMP + low ATP
- D. High ADP + high fructose-2,6-bisphosphate + low citrate
✅ Answer: B. High ATP + high citrate + low fructose-2,6-bisphosphate
Why correct: PFK-1 is inhibited by ATP (high energy state), citrate (TCA cycle running well → enough acetyl-CoA, no need for more pyruvate), and low F-2,6-BP (the most potent activator is absent). All three act simultaneously to switch off glycolysis when the cell is energy-replete.
Why A is wrong: High AMP activates PFK-1 (low energy signal), and high F-2,6-BP is the strongest activator — this combination would maximally stimulate glycolysis, not inhibit it.
Why C is wrong: High insulin raises F-2,6-BP (which activates PFK-1), and high AMP + low ATP are low-energy signals that activate glycolysis.
Why D is wrong: High ADP and high F-2,6-BP are both activators of PFK-1; low citrate removes an inhibitor — this combination stimulates glycolysis.
Exam tip: When a question says “aerobic, well-fed, hepatocyte” — translate that to: high ATP, high citrate, glucagon low/insulin high… then determine the net effect on each enzyme.
Q4: A 3-year-old child presents with chronic haemolytic anaemia, jaundice, and splenomegaly. The Coombs test is negative. Peripheral smear shows echinocytes. Red blood cell 2,3-BPG levels are markedly elevated. Which enzyme deficiency is responsible?
- A. Glucose-6-phosphate dehydrogenase
- B. Pyruvate kinase
- C. Triosephosphate isomerase
- D. Phosphoglycerate kinase
✅ Answer: B. Pyruvate kinase
Why correct: PK deficiency blocks Step 10 → 1,3-BPG accumulates → shunted into the Rapoport-Luebering pathway → excess 2,3-BPG. RBCs lose ATP → membrane failure → haemolysis. Morphology shows echinocytes (spiculated cells), not spherocytes. Coombs-negative confirms it is not immune-mediated.
Why A is wrong: G6PD deficiency causes episodic (not chronic) haemolysis triggered by oxidants (drugs, infections), with Heinz bodies on smear. 2,3-BPG is not specifically elevated.
Why C is wrong: TPI deficiency also causes haemolytic anaemia, but its hallmark is the addition of progressive neuromuscular disease — not mentioned here.
Why D is wrong: PGK deficiency is very rare and can cause haemolysis + neurological features, but elevated 2,3-BPG is the specific pointer to PK deficiency.
Exam tip: Elevated 2,3-BPG + Coombs-negative haemolytic anaemia + echinocytes = PK deficiency. This triad is tested repeatedly. Memorise it.
Q5: A researcher blocks GAPDH activity with iodoacetate in an RBC suspension and then adds arsenate. Compared to a control RBC suspension with glucose only, what is the most likely outcome in the experimental tube?
- A. Complete cessation of glucose consumption
- B. Normal glucose consumption with normal ATP production
- C. Continued glucose consumption up to Step 5 only, with accumulation of triose phosphates
- D. Glucose consumption continues past Step 6 but no ATP is produced from substrate-level phosphorylation
✅ Answer: C. Continued glucose consumption up to Step 5 only, with accumulation of triose phosphates
Why correct: Iodoacetate irreversibly inhibits GAPDH (Step 6). Glycolysis can proceed through Steps 1–5 (the investment phase), producing DHAP and G3P — but cannot proceed past Step 6. Arsenate in this context cannot even exert its effect because the GAPDH active site is already blocked. Triose phosphates (DHAP and G3P) accumulate.
Why A is wrong: Glycolysis is not completely stopped — Steps 1–5 still run. Only Steps 6 onwards are blocked.
Why B is wrong: With GAPDH blocked, there is no substrate for Steps 7–10, so no ATP can be generated in the payoff phase.
Why D is wrong: Option D describes arsenate poisoning alone (glycolysis runs past Step 6 but no ATP is made). With iodoacetate added, glycolysis cannot even get past Step 6.
Exam tip: When two inhibitors are combined in an experimental question, identify which one acts first in the pathway — that one determines the effective block point. Iodoacetate blocks Step 6; arsenate also acts at Step 6 but requires GAPDH to be functional. GAPDH is destroyed by iodoacetate first.
📚 References
📖 Harper’s Illustrated Biochemistry — Rodwell et al. | Chapter 17: The Glycolytic Pathway
📖 Lippincott’s Illustrated Reviews: Biochemistry — Ferrier | Chapter 8: Introduction to Carbohydrate Metabolism
📖 Lehninger Principles of Biochemistry — Nelson & Cox | Chapter 14: Glycolysis, Gluconeogenesis, and the Pentose Phosphate Pathway
📖 Stryer’s Biochemistry — Berg, Tymoczko & Stryer | Chapter 16: Glycolysis and Gluconeogenesis
📖 Vasudevan & Sreekumari’s Textbook of Biochemistry — Vasudevan | Chapter 6: Metabolism of Carbohydrates
🚀 Keep Practising — You Are Not Done Yet
Knowing the theory is only half the battle. The other half is applying it under exam pressure.
medicalmcq.in has free Biochemistry MCQs — every single one with a detailed explanation exactly like this article.
Work through them topic by topic, just like you read this guide, and you will walk into your exam knowing this subject cold.
When you are ready to simulate real exam conditions, our Mock Test Series gives you timed, subject-wise and full-length papers with performance tracking — so you always know exactly where to focus next.